9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) 3CO(g) +...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
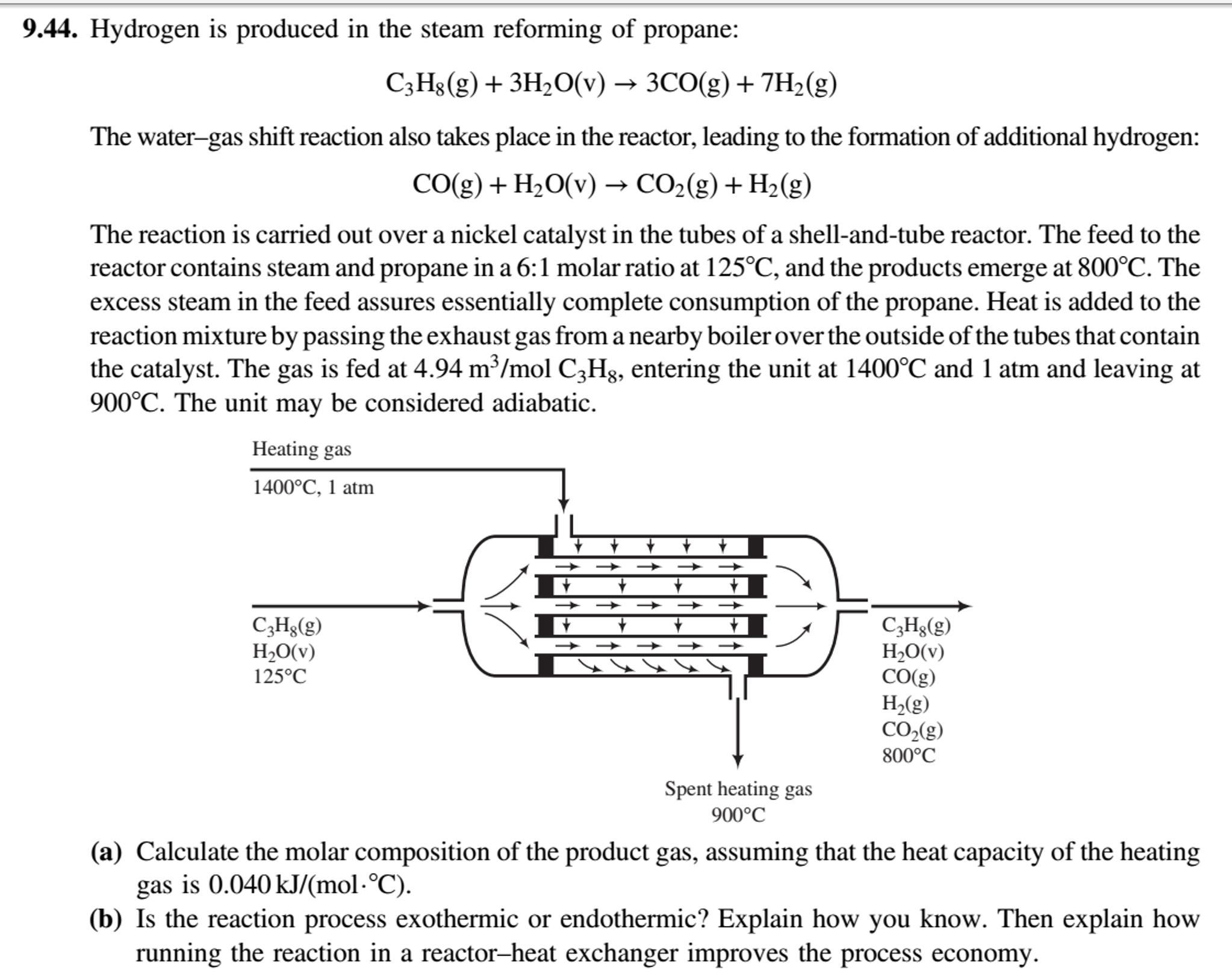
9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) → 3CO(g) + 7H2(g) The water-gas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: CO(g) + H2O(v) → CO2(g) + H2(g) The reaction is carried out over a nickel catalyst in the tubes of a shell-and-tube reactor. The feed to the reactor contains steam and propane in a 6:1 molar ratio at 125°C, and the products emerge at 800°C. The excess steam in the feed assures essentially complete consumption of the propane. Heat is added to the reaction mixture by passing the exhaust gas from a nearby boiler over the outside of the tubes that contain the catalyst. The gas is fed at 4.94 m³/mol C3H3, entering the unit at 1400°C and 1 atm and leaving at 900°C. The unit may be considered adiabatic. Heating gas 1400°C, 1 atm C3Hg(g) H2O(v) 125°C C,Hg(g) H,O(v) CO(g) H2(g) CO,(g) 800°C Spent heating gas 900°C (a) Calculate the molar composition of the product gas, assuming that the heat capacity of the heating gas is 0.040 kJ/(mol·°C). (b) Is the reaction process exothermic or endothermic? Explain how you know. Then explain how running the reaction in a reactor–heat exchanger improves the process economy. 9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) → 3CO(g) + 7H2(g) The water-gas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: CO(g) + H2O(v) → CO2(g) + H2(g) The reaction is carried out over a nickel catalyst in the tubes of a shell-and-tube reactor. The feed to the reactor contains steam and propane in a 6:1 molar ratio at 125°C, and the products emerge at 800°C. The excess steam in the feed assures essentially complete consumption of the propane. Heat is added to the reaction mixture by passing the exhaust gas from a nearby boiler over the outside of the tubes that contain the catalyst. The gas is fed at 4.94 m³/mol C3H3, entering the unit at 1400°C and 1 atm and leaving at 900°C. The unit may be considered adiabatic. Heating gas 1400°C, 1 atm C3Hg(g) H2O(v) 125°C C,Hg(g) H,O(v) CO(g) H2(g) CO,(g) 800°C Spent heating gas 900°C (a) Calculate the molar composition of the product gas, assuming that the heat capacity of the heating gas is 0.040 kJ/(mol·°C). (b) Is the reaction process exothermic or endothermic? Explain how you know. Then explain how running the reaction in a reactor–heat exchanger improves the process economy.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following reaction is carried out at 500 K in a container equipped with a movable piston. After the reaction has reached equilibrium, the container has the composition depicted here. Suppose the...
-
If the reaction is carried out at constant V, how does the total pressure change if T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
The following elementary homogeneous gas-phase reaction is carried out isothermally in an ideal, steady state 0.2 L plug flow reactor: 2 F --> P. PThe inlet molar feed consists of a 50%-50% mixture...
-
On May 1, Johnson Corporation purchased inventory for $40,000 on credit. On May 15, Johnson sold inventory with a cost of $10,000 for $25,000 on credit. Prepare journal entries to record these...
-
A number of nutritionists have argued that fastfood restaurants have a negative effect on nutrition quality. In this exercise you are asked to determine if there is evidence to conclude that...
-
Nitric oxide (NO) emissions from automobile exhaust can be reduced by using a catalytic converter, and the following reaction occurs at the catalytic surface: \[\mathrm{NO}+\mathrm{CO} ightarrow...
-
Yamaguchi Company makes printed circuit boards in a suburb of Kyoto. The production process is automated with computer-controlled robotic machines assembling each circuit board from a supply of...
-
At the end of 2014, Frontier Corporation has $360,000 of cumulative temporary differences that will result in reporting future taxable amounts as follows. 2015 ......... $105,000 2016 .........90,000...
-
Say No to the Brush, LLC is a manufacturer of premium hairstyling products. Its most popular product, Its Real, is sold in 10 ounce bottles. One bottle of Its Real is expected to use 8 ounces of...
-
1. What is your assessment of the financial performance of Nelson Nurseries? 2. Do you agree with Christine Barton?s accounts-payable policy? 3. What explains the erosion of the cash balance? 4. What...
-
Question 21 Which of the following can be implemented by using the basic summing amplifier? Not yet answered Select one: Marked out of O a Scaling amplifier O b. Averaging amplifier O c. All of the...
-
Consider the following pool of mortgages: 100 mortgages with initial balance of $118,580, interest rate 4.7%, issued for 30 years with monthly payments 80 mortgages with initial balance of $354,365,...
-
Write the program in c + + ?using include iostream and namespace and do not use global varients. Explain your code and use comments and write the code in simple terms to understand and easy to read....
-
Providing mental health services is an important component of any effort to reduce gun violence in a healthcare facility, what are the prevention the facility can take in order to reduce gun violence...
-
The last thing you need is the values of penalty you are trying. This can be created using grid _ regular ( ) which creates a grid of evenly spaces parameter values. You use the penalty ( ) function...
-
In your project, explain your codes with comments. Without comments, your project will not be marked. This Project needs to be written in Java and on Android Studio Problem In this Project you are...
-
Pablo Company is considering buying a machine that will yield income of $3,300 and net cash flow of $18,200 per year for three years. The machine costs $55,500 and has an estimated $10,800 salvage...
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
List two applications of freezing-point depression.
-
Why is it generally impossible to predict the rate law for a reaction on the basis of the chemical equation only?
-
Photographers hypo, used to fix negatives during the development process, is sodium thiosulfate pentahydrate. What is the chemical formula of this compound?
-
What are the key elements of working capital?
-
From the extracts of data provided below for Easy Events, calculate the ratios the following ratios and explain what they reveal. a) ROCE b) Asset utilisation c) Net profit percentage d) Gross profit...
-
Name the three working capital policies a business can follow.

Study smarter with the SolutionInn App


