Answered step by step
Verified Expert Solution
Question
1 Approved Answer
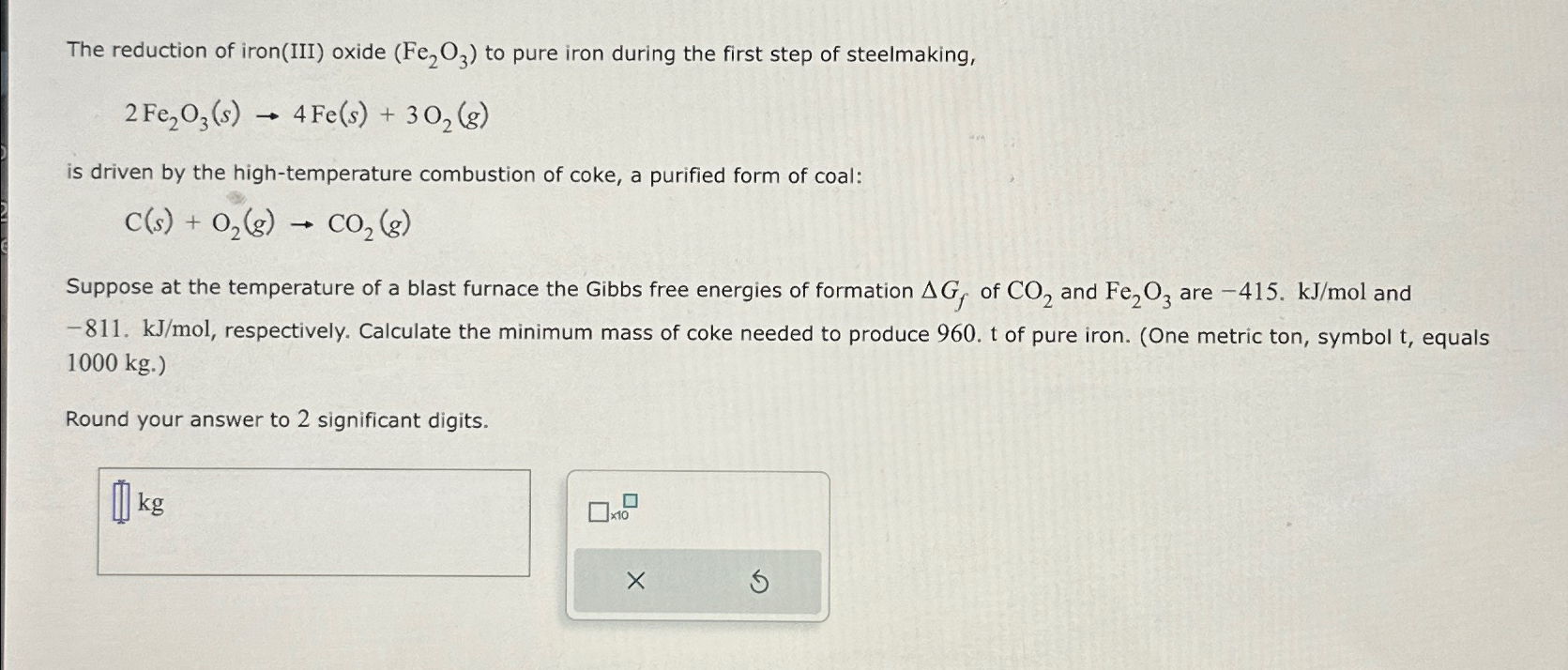
The reduction of iron ( III ) oxide ( F e 2 O 3 ) to pure iron during the first step of steelmaking, 2
The reduction of ironIII oxide to pure iron during the first step of steelmaking,
is driven by the hightemperature combustion of coke, a purified form of coal:
Suppose at the temperature of a blast furnace the Gibbs free energies of formation of and are and respectively. Calculate the minimum mass of coke needed to produce of pure iron. One metric ton, symbol equals
Round your answer to significant digits.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


