Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The reform reaction between steam and gaseous methane (CH) produces synthesis gas, a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one
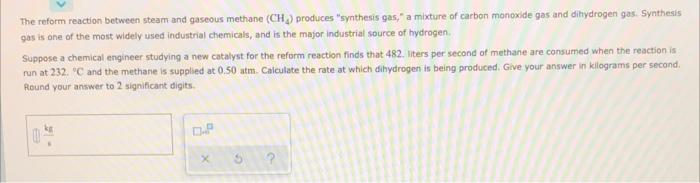 The reform reaction between steam and gaseous methane (CH) produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen Suppose a chemical engineer studying a new catalyst for the reform reaction finds that 482. iters per second of methane are consumed when the reaction is run at 232. C and the methane is supplied at 0.50 atm. Calculate the rate at which dihydrogen is being produced. Give your answer in klograms per second. Round your answer to 2 significant digits X
The reform reaction between steam and gaseous methane (CH) produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen Suppose a chemical engineer studying a new catalyst for the reform reaction finds that 482. iters per second of methane are consumed when the reaction is run at 232. C and the methane is supplied at 0.50 atm. Calculate the rate at which dihydrogen is being produced. Give your answer in klograms per second. Round your answer to 2 significant digits X

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


