Answered step by step
Verified Expert Solution
Question
1 Approved Answer
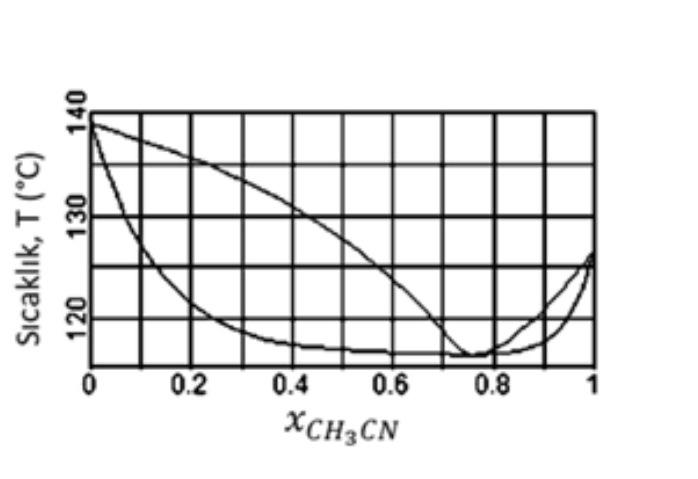
The temperature component graph formed by the binary mixture of Water - Acetonitrile under 3,5 bar pressure is as on the side. By using this
The temperature component graph formed by the binary mixture of Water - Acetonitrile under 3,5 bar pressure is as on the side. By using this chart; Temperature, T ("C)
A-) When the concentration of acetonitrile (CH,CN) mixed with water is analyzed, it is observed that acetonitrile has 0,43 mole fraction in this mixture. What is the approximate mole fraction of acetonitrile in the distillate (in the deposited distillate) when distilled by simple (ordinary) distillation?
B-) If 100ml of water and 100ml of acetonitrile are mixed under the same conditions, would the volume of the mixture be 200ml, more or less?
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


