Question
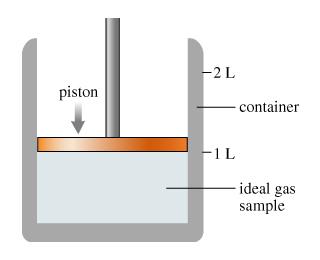
This figure (Figure 1)shows a container that is sealed at the top by a movable piston. Inside the container is an ideal gas at 1.01
This figure (Figure 1)shows a container that is sealed at the top by a movable piston. Inside the container is an ideal gas at 1.01 bar, 20.0 C, and 1.00 L. This information will apply to all parts of this problem A, B, and C. Figure1 of 1 There is a two-litres container with a piston filled with one litre of an ideal gas sample. Part A What will the pressure inside the container become if the piston is moved to the 1.20 L mark while the temperature of the gas is kept constant? Express your answer with the appropriate units. Part B The gas sample has now returned to its original state of 1.01 bar, 20.0 C and 1.00 L. What will the pressure become if the temperature of the gas is raised to 200.0 C and the piston is not allowed to move? Express your answer with the appropriate units. Part C The gas described in parts A and B has a mass of 1.66 grams. The sample is most likely which monoatomic gas? Type the elemental symbol of the gas below.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


