Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Use the balanced equation to answer the following question. 6CuNO3 + Al2(SO4)3 Molar mass of CuNO3 3Cu2SO4 + 2Al(NO3)3 125.56 g/mol 213.01 g/mol Molar
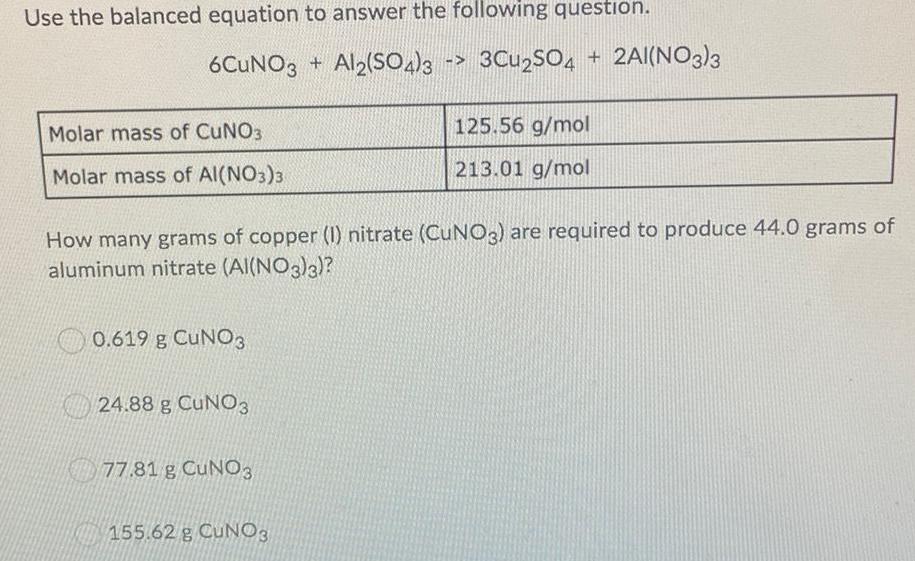
Use the balanced equation to answer the following question. 6CuNO3 + Al2(SO4)3 Molar mass of CuNO3 3Cu2SO4 + 2Al(NO3)3 125.56 g/mol 213.01 g/mol Molar mass of Al(NO3)3 How many grams of copper (I) nitrate (CuNO3) are required to produce 44.0 grams of aluminum nitrate (Al(NO3)3)? 0.619 g CuNO3 24.88 g CuNO3 77.81 g CuNO3 155.62 g CUNO3
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


