Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Use the References to access important values if needed for this question. The following thermochemical equation is for the reaction of carbon monoxide() with hydrogen()
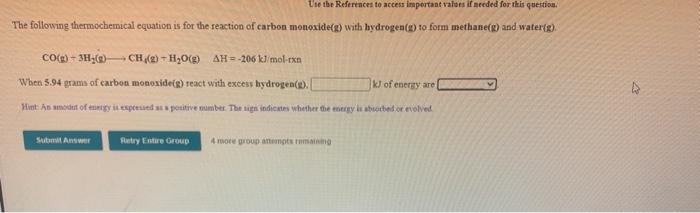 Use the References to access important values if needed for this question. The following thermochemical equation is for the reaction of carbon monoxide() with hydrogen() to form methanes) and waterig). CO(g) + 3HC) CH (2) - H2O(9) AH=-206 tel mol-exa When 5.94 grams of carbon monoxide(s) react with excess hydrogen. k) of energy are Hint: An modest of trgy exposed a positive number. The sign indicates whether the energy le abecebed or evolved Suomen Retry Entire Group 4 more group angin
Use the References to access important values if needed for this question. The following thermochemical equation is for the reaction of carbon monoxide() with hydrogen() to form methanes) and waterig). CO(g) + 3HC) CH (2) - H2O(9) AH=-206 tel mol-exa When 5.94 grams of carbon monoxide(s) react with excess hydrogen. k) of energy are Hint: An modest of trgy exposed a positive number. The sign indicates whether the energy le abecebed or evolved Suomen Retry Entire Group 4 more group angin

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


