What valence orbitals, if any, remain unhybridized on the O atom in Cl O? Drag the...
Fantastic news! We've Found the answer you've been seeking!
Question:
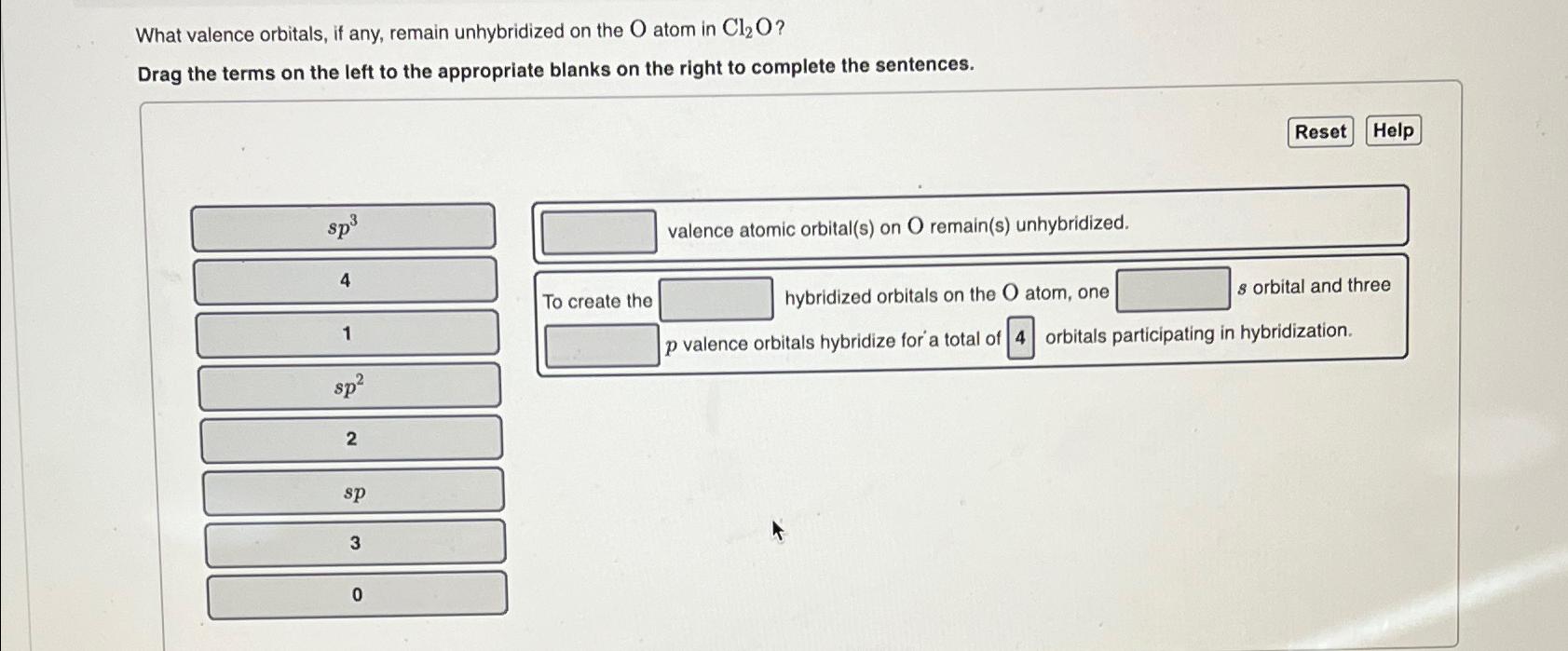
Transcribed Image Text:
What valence orbitals, if any, remain unhybridized on the O atom in Cl O? Drag the terms on the left to the appropriate blanks on the right to complete the sentences. sp 4 1 sp 2 sp 3 0 To create the valence atomic orbital(s) on O remain(s) unhybridized. Reset Help s orbital and three hybridized orbitals on the O atom, one p valence orbitals hybridize for a total of 4 orbitals participating in hybridization. What valence orbitals, if any, remain unhybridized on the O atom in Cl O? Drag the terms on the left to the appropriate blanks on the right to complete the sentences. sp 4 1 sp 2 sp 3 0 To create the valence atomic orbital(s) on O remain(s) unhybridized. Reset Help s orbital and three hybridized orbitals on the O atom, one p valence orbitals hybridize for a total of 4 orbitals participating in hybridization.
Expert Answer:
Answer rating: 100% (QA)
Solutions Step 1 Oxygen has the electron configuration 1s22s22p4 In the process of forming bonds oxygen will use its valence electrons which are in th... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
The accompanying data table shows reaction distances in centimeters for the dominant hand for a random sample of 40 independently chosen college students. Smaller distances indicate quicker...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
After the Internet investment bubble burst, the original concept of easyInternetcaf? (eIc) was just not working out. The management decided to retrench, closing and downsizing some of the original...
-
Find the magnitude of the reaction at A and the tension in cable CD using (a) the con- cepts of three-force bodies (closed polygon method discussed in chapter 2). (b) Check your answers by doing sum...
-
Using the Muskingum method, route the following inflow hydrograph assuming K = 4 hr, x = 0.1, Plot the inflow and outflow hydro graphs for each case assuming initial outflow equals initial inflow....
-
Pennington Hardwood Lumber Supplier has three lumber mills in San Jose, Dayton, and Boston that manufacture pre-cut hardwood. These pre-cut hardwood are shipped to building warehouses in Cleveland,...
-
Effects of Discounts on Sales and Purchases} Smithson Products sells shoes and accessories to retail stores. Gross sales in 2018 were \(\$ 1,500,250\) (Smithson's list price) on terms \(4 / 10,...
-
The management of Dorsch Aluminum Co. is considering whether to process aluminum ingot further into rolled aluminum. Rolled aluminum can be sold for $4,100 per ton, and ingot can be sold without...
-
A firm is considering replacing a machine that has been used to make a certain kind of packaging material. The new, improved machine will cost $31,000 installed and will have an estimated economic...
-
You work in the human resources department of your company helping new employees fill out the necessary paperwork to get their first paycheck. There are a number of decisions that employees must make...
-
Required information [The following information applies to the questions displayed below.] Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company...
-
Lab 27 Network Management with ipMonitor Part 1: Install ipMonitor 1. ipMonitor is a free network monitoring software which identifies SNMP agents on a network. Click the link to go to the download...
-
The following code computes the intersection (the number of elements in common) of two arrays. It assumes that neither array has duplicates. It computes the intersection by sorting one array (array...
-
1. Assume a nucleophile displaced a leaving group in both the alcohol and the protonated alcohol shown below. Using Nue for the nucleophile, draw curved arrows to show the formation of the product...
-
A seashore sand mixture contains 85% insoluble sand, 12% salt, and 3% water by weight. 100 kilograms of this mixture is to be washed by batch operations so that it will contain less than 0.2% salt...
-
Ununununununununununununununun 3. A binary transmission system uses the following waveform and its antipodal counterpart to represent binary 1 and 0 symbols respectively. On the receiver, a...
-
Exercise 11-30 Reconstruct Missing Informa- tion from Partial Data (LO 11-2, 11-5) You brought your work home one evening, and your nephew spilled his chocolate milk shake on the variance report you...
-
Funds are separate fiscal and accounting entities, each with its own self-balancing set of accounts. The newly established Society for Ethical Teachings maintains two funds-a general fund for...
-
Emily Jackson (Social Security number 765-12-4326) and James Stewart (Social Security number 466-74-9932) are partners in a partnership that owns and operates a barber shop. The partnership's first...
-
Janie graduates from high school in 2012 and enrolls in college in the fall. Her parents pay $4,000 for her tuition and fees. a. Assuming Janie's parents have AGI of $170,000, what is the American...
-
Matthew borrows $250,000 to invest in bonds. During 2012, his interest on the loan is $30,000. Matthew's interest income from the bonds is $10,000. This is Matthew's only investment income. a....
-
Continuation of S10-5: cash budget (Learning Objective 3) Refer to S10-5. Grippers has $8,300 cash on hand on January 1. The company requires a minimum cash balance of $7,500. January cash...
-
Revise cash payments for purchases (Learning Objective 4) Refer to the original Whitewater Sporting Goods Data Set item 5. Suppose Whitewater Sporting Goods pays for 60% of inventory purchases in the...
-
Revise cash collections budget (Learning Objective 4) Turn to the original Whitewater Sporting Goods Data Set item 4. Suppose 70% of sales are cash and 30% are credit. Revise Whitewater's sales...

Study smarter with the SolutionInn App


