Answered step by step
Verified Expert Solution
Question
1 Approved Answer
() X STARTING AMOUNT How many moles of Na SO are in a 0.140 m aqueous solution that was prepared with 2.30 kg of water?
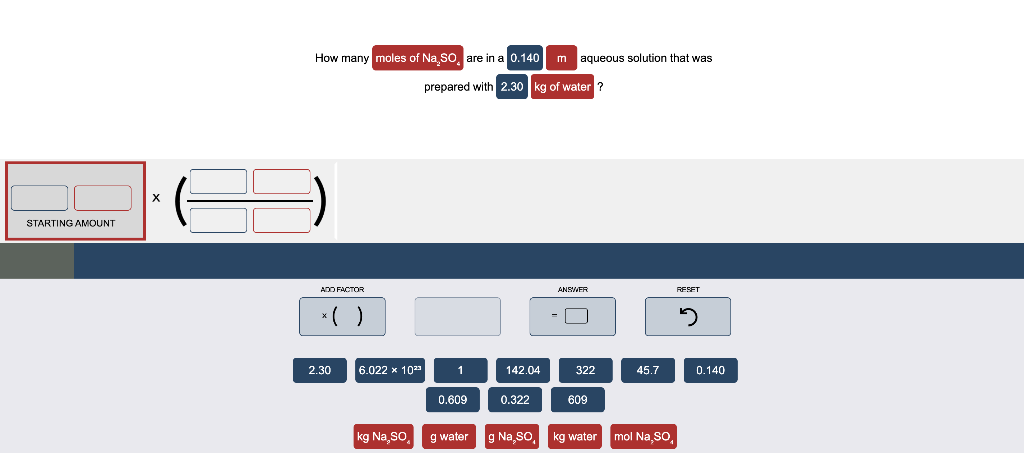
X STARTING AMOUNT How many moles of Na SO are in a 0.140 m aqueous solution that was prepared with 2.30 kg of water? ADO FACTOR .( ) ANSWER RESET 2.30 6.022 x 1023 1 142.04 322 45.7 0.140 0.609 0.322 609 kg Na,SO g water g Na,SO kg water mol Na,SO
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


