Arrange the following three chlorides in decreasing order of S N 1 reactivity. (a) A > B
Question:
Arrange the following three chlorides in decreasing order of SN1 reactivity.
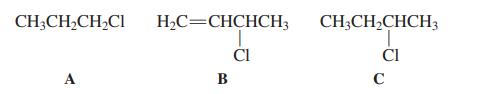
(a) A > B > C; (b) B > C > A; (c) B > A > C; (d) C > B > A.
Transcribed Image Text:
CH;CH,CH2CI H2C=CHCHCH3 CH;CH,CHCH3 ČI ČI A B C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
The correct answer of this question is option b ie B C A ie Arrangement of following th...View the full answer

Answered By

Aditi Deswal
Currently , I am doing post graduation ( MSc.) in Chemistry . I want to bacome a lecturer . I feel happy when I teach student and solve their problem . It is my passion as well as hobby . I feel blessed If I got a chance to share my knowledge as much as I have . I teach student at my home also. I love teching and want to spend my whole life to teach the students and explores new things.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Arrange the following compounds in order of decreasing SN2 reactivity toward sodium ethoxide: CH3 CH3 CH3CH2CHB CH CHCH2Br CH,CH2CH2CH2Br
-
Rank the following compounds in decreasing order of their reactivity toward the SN2 reaction with sodium ethoxide (Na+ Ì OCH2CH3) in ethanol. methyl chloride isopropyl bromide tert-butyl...
-
(a) Rank ethanol, methylamine, and acetic acid in decreasing order of acidity. (b) Rank ethanol, methylamine (pKb 3.36), and ethoxide ion (CH3CH2O-) in decreasing order of basicity. In each case,...
-
Harpo Marks Corporation produces and sells forklift trucks. Model 17A (the Chico) has a list price of $ 70,000. The production cost for the 20 finished Chicos in inventory at the end of 20X4 was $...
-
As a student, you generally take more than one course during a given semester. Each course requires different amounts of your time, at varying schedules. Many students spend more time on the courses...
-
If you are the director of crisis management in an international institution, mention the following: 1- What crisis has your organization been exposed to? 2- What is The vision and mission of your...
-
List some qualities of a productive board meeting.
-
City Segway Tours of Washington DC, LLC (CST), operated tours where customers used Segway personal transportation vehicle to tour the city. Norman Mero and his signifi-cant other signed up for such a...
-
Coca-Cola decided to market all of its top beverages under one tagline. What is the main goal behind this move? Consumers are less likely to confuse the many beverages if there is a single tagline. A...
-
The objective of this exercise is to develop your ability to perform a comprehensive analysis on a set of financial statements. Use the copy of the 2018 annual report of Dollarama Inc. (year end...
-
How many nodes are present in the LUMO (lowest unoccupied molecular orbital) of 1,3- butadiene? (a) Zero; (b) one; (c) two; (d) three; (e) four
-
When cyclopentadiene is treated with tetracyanoethene, a new product results. Its most likely structure is CN CN CN () - CN CN (b) N N CN CN CN H,C-C-CN () H2C-C-CN (d) CN NC CN CN
-
The accounts below appear in the ledger of Popovich Company. Instructions From the postings in the accounts above, indicate how the information is reported on a statement of cash flows by preparing a...
-
Define "corroborative" evidence.
-
Which of the following models expresses the general relationship of risks associated with the auditor's evaluation of control risk (C R), inherent risk (I R), and audit risk (A R) that would lead the...
-
Which of the following combinations of engagement risk, audit risk, and mate- riality would lead to the most audit work? Engagement Risk a. Low b. Moderate c. Low d. High Audit Risk High Low Moderate...
-
How does direct evidence differ from hearsay evidence in a courtroom?
-
Is evidence that warrants "no reliance" of any use? Explain your response.
-
The following table lists diffraction angles for the first three peaks (first-order) of the x-ray diffraction pattern for some metal. Monochromatic x-radiation having a wavelength of 0.0711 nm was...
-
How is use of the word consistent helpful in fraud reports?
-
The first electronically excited state of O is Ill. and lies 7918.1 cm-1 above the ground state, which is Calculate the electronic contribution to the molar Gibbs energy of 0, at 400 K.
-
Estimate the contribution of the spin to the molar entropy of a solid sample of a d-metal complex with S =3/2
-
Calculate the value of K at 298 K for the gas-phase isotopic exchange reaction 2 79Br81Br79Br79 ( 79Br79Br+ 81Br81Br the Br2 molecule has a non-degenerate ground state, with no other electronic...
-
Table below contains the percentage of woman receiving prenatal care in 2009 for a random sample of countries ('Pregnant woman receiving,' 2013). Estimate the mean percentage of woman receiving...
-
Problem #5 You still $10,000 to pay for a truck. You know you need 18.6% interest to get you there. You can't find that rate anywhere. The best you can find is an annual rate of 13.13%. How long will...
-
The following is a lease for a doughnut-making machine: Annual payment (due at commencement date) $22,000 Lease term 5 Incremental borrowing rate 10% Implicit rate (not readily determinable by...

Study smarter with the SolutionInn App


