Formulate the expected product of each of the following reactions. H;CO. H,C CH3 hv hv () (b)
Question:
Formulate the expected product of each of the following reactions.
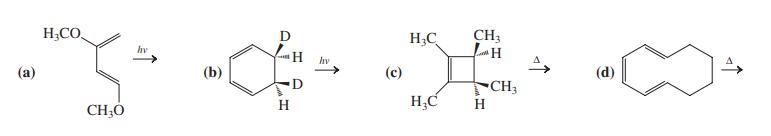
Transcribed Image Text:
H;CO. H,C CH3 hv hv (а) (b) (с) (d) D CH3 H H;C H. CH;Ó
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a b c d OCH ...View the full answer

Answered By

Deepak Kumar
In the teaching profession, one thing I truly believe is that " to be a teacher you don't only need knowledge of the subject but you have the ability and skill to make the student understand". I have taught in summer school programs two times which is organized by the Society for Promotion of Science and Technology in India( SPSTI).
and I have done a school internship for one semester. I love to teach.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the expected product of each of the following reaction sequences. (a) (b) (c) H 1. LIAIH,, (CH,CH,),O 2. H., . H
-
Formulate the potential product of each of the following reactions. As you did in Problem 44, write "no reaction" where appropriate. (Identify the expected leaving group in each of the substrates and...
-
Formulate the product of each of the following reactions. The solvent in each case is (CH3CH2)2O. (a) (b) (c) O .LiAID 2. H, H,O CH3CH O 1.LIAIH 2D D.O LiAID CH,CH>
-
Another equation that has been used to model population growth is the Gompertz equation: dy dt ky In(K/y) , where k and K are positive constants. For each y in 0 < y K, show that dy/ dt is never less...
-
How does the real options approach apply to the selection of IT projects?
-
Do 10 steps. Solve exactly. Compute the error. Show details. y' = (y - x) 2 , y(0) = 0, h = 0.1
-
To examine the nature of interviewing in grievance and discipline?
-
Kidstuff, Inc., was started 10 years ago by selling childrens clothing through catalogs. Helena Cence, Kidstuffs controller, had determined that the cost of filling and shipping orders was fairly...
-
Give example for the graph of NPV vs IRR concave downward and upward(multiple IRR). explain when will happen that two conditions.
-
Morelli Electric Motor Corporation manufactures electric motors for commercial use. The company produces three models, designated as standard, deluxe, and heavy-duty. The company uses a job-order...
-
Bicyclic diene A reacts readily with appropriate alkenes by the Diels-Alder reaction, whereas diene B is totally unreactive. Explain. A B
-
Which of the reactions shown below will occur under the influence of heat? Light? (a) (b) (c) H H
-
Determine the vertical displacement of joint D. Using Castiglianos theorem. AE is constant. Assume the members are pin connected at their ends. 000 4 m 4 m - 15 kN 20 kN 000
-
A liquid hydrocarbon mixture was made by adding 295 kg of benzene, 289 kg of toluene and 287 kg of p-xylene. Assume there is no change of volume upon mixing, i.e., Vmix=0 , in order to determine: 1....
-
b) Maseru Development Bank has R850 million credit with Matsieng Hydroelectric Power, with a maturity of eighteen months. The expected loss for Maseru Development Bank is R22 million, and the...
-
Assume an organic compound has a partition coefficient between water and ethyl acetate equal to 8.12. If there are initially 7.10 grams of the compound dissolved in 75.0 mL of water, how many grams...
-
Berger Paint Pakistan limited produces three types of joint products, S ilver paint, G olden paint and D iamond paint. During March, 2020, the following information was recorded : Particulars Silver...
-
Choose an industry in which you are interested in working. How is that industry trending? What internal and external factors may affect the direction of the organization? For your initial post,...
-
Write a parameterized class definition for a doubly linked list that has a parameter for the type of data stored in a node. Make the node class an inner class. Choosing which methods to define is...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
What is the temperature of a two-level system of energy separation equivalent to 300 cm-I when the population of the upper state is one-half that of the lower state?
-
Calculate the ratio of the translational partition functions of xenon and helium at the same temperature and volume.
-
A certain atom has a doubly degenerate ground level, a triply degenerate electronically excited level at 1250 cm3, and a doubly degenerate level at 1300 cm3. Calculate the partition function of these...
-
How do external factors such as changing consumer preferences affect the retail industry?"
-
Please show workings :) Oxford Company has limited funds available for investment and must ration the funds among four competing projects. Selected information on the four projects follows: Life of...
-
At a 3% (EAR) rate of interest, you will quadruple (increase four folds) your money in approximately ____ years.

Study smarter with the SolutionInn App


