Give the expected major product of addition of each of the following reagent mixtures to benzene. (a)
Question:
Give the expected major product of addition of each of the following reagent mixtures to benzene.
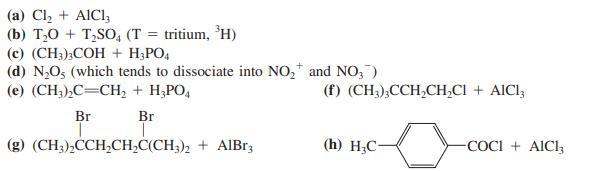
Transcribed Image Text:
(a) Cl, + AICI, (b) T,0 + T,SO, (T = tritium, 'H) (с) (CH).СОН + Н.РО (d) N,O; (which tends to dissociate into NO, and NO, ) (е) (CH,),С—СН, + Н.РО, (f) (CH3);CCH,CH,CI + AICI, Br Br (g) (CH;),CCH,CH,C(CH;), + AlBr3 (h) H;C- COCI + AICI3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a C C AICI c 656 Cl b Same Preci line CCH33 d NO5 ...View the full answer

Answered By

Sk Bappa
Styding msc in chemistry and also aspirant of csir net and gate exam. Also I teach chemistry as a teacher
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the expected major product of each of the following reactions. (a) (b) (c) (d) CH CH,CH,OH COnC HI Conc. HBr (CH,)CHCH.CH.OH- Conc. HI OH CH,CH2,COH COn HCT
-
Give the expected major product of each of the following reactions. PCC is the abbreviation for pyridinium chlorochromate (Section 8-6). (a) (b) (c) (d) (e) CH CH CH,OH NeCrO HSO, HO PCC, CH,CI (CH3)...
-
Give the expected major product of each of the following reaction sequences. PCC refers to pyridinium chlorochromate. (a) (b) (c) 1. CrO, H,SO., HO 2. CH.CHMgBr, (CH,CH, 3. H, H,O CHs) CHOH 1. OH,...
-
in an armicie entitled Fuel Economy Calculations tio Be Alteted lames Healey indicated that the goermnt planned to change how it caleulates fuel economy for new cas and trucks Thia in the first...
-
Write a brief explanation of each of the following PDLC methods and explain the strengths and weaknesses of each: waterfall, iterative and incremental, spiral, Scrum, XP, and RUP.
-
Did the actual spending stay within the projected budget? If not, where did the overspending occur? Be sure to consider those planned expenses vs impulse expenses. Was the proposed budget appropriate...
-
We wish to strip \(\mathrm{SO}_{2}\) from water using pure air at \(20.0^{\circ} \mathrm{C}\). Outlet water contains \(0.0060 \mathrm{~mol} \% \mathrm{SO}_{2}\), and inlet water contains \(0.112...
-
Northwest Paperboard Company, a paper and allied products manufacturer, was seeking to gain a foothold in Canada. Toward that end, the company bought 40% of the outstanding common shares of Vancouver...
-
In two to three paragraphs, answer the following questions. i need brief answer with 50-100 words How is social intelligence related to risk? Does social intelligence help with risk assessment and...
-
Three former RIM employees decided to go into business for themselves and open a store near an office park to sell wireless equipment to young professionals. Their first products were cell phones,...
-
Reaction review. Without consulting the Reaction Road Map on p. 721, suggest a reagent to convert benzene into each of the following compounds. C(CH3)3 NO2 CH3 CH,CH; SO,H Br (e) (f) (h)
-
Write mechanisms for reactions (c) and (f) in Problem 48. Data from Problem 48 Give the expected major product of addition of each of the following reagent mixtures to benzene. (a) Cl, + AICI, (b)...
-
Find the equation of the least-squares line for the given data. Graph the line and data points on the same graph. 20 26 30 38 48 60 y 160 145 135 120 100 90 X
-
The auditors for a chain of wholesale food distribution outlets conducted an audit of the companys produce management. They discovered what appeared to be high levels of spoilage in the three oldest...
-
What is The Institute of Internal Auditors? Discuss briefly its history and current functions.
-
Explain the three primary responsibilities of management. How does internal auditing relate to each of these management responsibilities?
-
A car's radio may be subjected to voltage spikes induced by coupling from the ignition system. Pulses on the order of \(\pm 250 \mathrm{~V}\) and lasting for \(120 \mu \mathrm{s}\) may exist. Design...
-
The auditors for Drawing Heavy Industries, Inc. recently completed an audit of the company-owned store used by its employees and their families. Following are the findings and recommendations...
-
Cobalt (Co) has an HCP crystal structure, an atomic radius of 0.1253 nm, and a c/a ratio of 1.623. Compute the volume of the unit cell for Co?
-
Explain why each of the following is either a private good or a public good: traffic lights, in line skates, a city park, a chicken salad sandwich, a tennis racket, national defense, a coastal...
-
Self-assembled monolayers (SAMs) are receiving more attention than Longmuir-Blodgett (LB) films as starting points for nanofabrication. How do SAMs differ from LB films and why are SAMs more useful...
-
Explain the physical origins of surface activity by surfactant molecules.
-
Calculate the number-average molar mass and the mass-average molar mass of a mixture of two polymers, one having M = 62 kg mol-1 and the other M = 78 kg mol-1, with their amounts (numbers of moles)...
-
Construct a probability distribution for the sum shown on the faces when two dices, each with 6 faces, are rolled
-
Introduction External Url 10.1 Two Population Means with Unknown Standard Deviations External Url 10.2 Two Population Means with Known Standard Deviations External Url 10.3 Comparing Two Independent...
-
Select any multinational company and explain the following based on Strategic Outsourcing: 1. Strategic reasons with valid outcomes for outsourcing manufacturing and services. 2. Benefits and...

Study smarter with the SolutionInn App


