Rank the following six molecules in approximate order of decreasing S N 1 reactivity and decreasing S
Question:
Rank the following six molecules in approximate order of decreasing SN1 reactivity and decreasing SN2 reactivity.
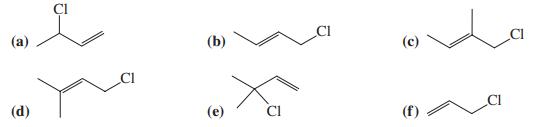
Transcribed Image Text:
(а) (b) CI (d) CI (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Reactivity order ...View the full answer

Answered By

Mohd Sahil
My self Mohd sahil having six years experience in teaching chemistry subject. Mentor of JEE and NEET aspirants. I know how to improve confidence and boost the energy level. Giving daily life examples so students easily understand the chapter.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Rank the following substances in order of their expected SN1 reactivity: Br Br H CHCCH CHCH2Br H2C=CHBr
-
Arrange the following compounds in order of decreasing SN2 reactivity toward sodium ethoxide: CH3 CH3 CH3CH2CHB CH CHCH2Br CH,CH2CH2CH2Br
-
Rank the following compounds in order of decreasing reactivity in an E2 reaction CH3 CH3 CH3 Br Br CH3
-
World Information Group has two major divisions: print and Internet. Summary financial data (in millions) for 2011 and 2012 are: The annual bonuses of the two division managers are based on division...
-
Explain the difference between a Theory X employee and a Theory Y employee.
-
Meredith Merriweather, CPA is the CFO of Trego Bikes and Trikes (TBT), a manufacturer of Bicycles ranging from tricycles to high-end racing bikes. The company has good market penetration and has seen...
-
Until relatively recently many employers in the UK were greatly restricted in their capacity to design reward strategies which met their priorities by the prevailing system of multi-employer or...
-
Whats next for Google? Is it doing the right thing taking on Microsoft with the concept of cloud computing, and Apple in the fight for smart phones?
-
In its IPO, 1 million company shares were issued at a price of $28 per share. The underwriter's fee for the offering was 10% of gross proceeds and the issuer incurred direct out-of-pocket costs of...
-
Information from Halloran Company's Income Statement and Balance Sheets for the years 2013-2017 is presented. HALLORAN COMPANY Comparative Income Statements For Years Ended December 31, 2017-2013 ($...
-
Rank the following six molecules in approximate order of decreasing SN1 reactivity and decreasing SN2 reactivity. () (b) CI (d) CI (f)
-
How would you expect the S N 2 reactivities of simple saturated primary, secondary, and tertiary chloroalkanes to compare with the S N 2 reactivities of the compounds in Problem 39? Make the same...
-
The barcode used by the U.S. Postal System to route mail is defined as follows: Each decimal digit in the ZIP code is encoded using a sequence of three half-height and two full-height bars. The...
-
Perform an analysis of Best Buy Co. Inc. Your analysis will draw on the Form 10K (as of February 2013). Your analysis can include information prior to February 2013 but should not draw on any...
-
Research organizational structure of a company of your choice. Use your understanding of organizational structure to analyze whether this organization's structure is the best choice for the business...
-
Tabletop Exercise (15%) Develop a tabletop exercise for your organization or community. The size and scope of your exercise can be whatever you need it to be in order for you to complete the...
-
Within the framework of the Porter Five-Forces Model of Competition, describe the competitive force of rivalry among competing sellers. What are some of the factors that increase the rivalry among...
-
The XYZ Corporation has decided to make some changes to help with the work-life balance of its employees. Currently, the organization has 40 employees: 25 full-time employees and 15 part-time...
-
Create a JavaFX application that will draw a fractal curve using line segments. Fractals are recursively defined curves. The curve you will draw is based on a line segment between points p and p : To...
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
In the group theoretical language developed in Chapter 12, a spherical rotor is a molecule that belongs to a cubic or icosahedral point group, a symmetric rotor is a molecule with at least a...
-
The protein haemerythrin is responsible for binding and carrying 0, in some invertebrates. Each protein molecule has two Fe'+ ions that are in very close proximity and work together to bind one...
-
The moments of inertia of the linear mercury (II) halides are very large, so the and S branches of their vibrational Raman spectra show little rotational structure. Nevertheless, the peaks of both...
-
question 1- You borrow a simple loan of SR 500,000, interest rate is 20%, it matures in one year. what's the yied to maturity? question 2- calculate_i for One-Year Discount Bond with price(p) =...
-
Five years ago, Diane secured a bank loan of $ 3 8 0 , 0 0 0 to help finance the purchase of a loft in the San Francisco Bay area. The term of the mortgage was 3 0 years, and the interest rate was 5...
-
Industry Current Year Minus 1 Current Year Minus 2 Company: Air Products and Chemicals, Inc. (APD) Stock Price: 306.72 USD Shares Outstanding: 220.89 M Financial Ratios Most Current Year Current...

Study smarter with the SolutionInn App


