Methanol is produced using syngas, a mixture (CO12 H2) containing CO2 as an inert impurity. Originally there
Question:
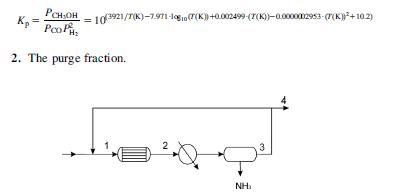
Methanol is produced using syngas, a mixture (CO12 H2) containing CO2 as an inert impurity. Originally there were 0.25 mol per 100 mol of syngas. The reactor operates at 25 bar and 232C. Assume that 90% of the methanol can be recovered condensing it. No water is produced since Co-based catalysts are used. The reactor can handle up to 5 moles of CO2 per 100 moles of syngas. Fig. P5.15 shows the scheme of the synthesis loop. Determine:
1. The reactor conversion assuming that the equilibrium constant of the process taking place in the reactor can be computed using the following equation:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Industrial Chemical Process Analysis And Design
ISBN: 9780081010938
1st Edition
Authors: Mariano Martín
Question Posted:





