A displacement reaction is an oxidation-reduction reaction in which one element displaces another from solution. In each
Question:
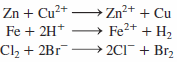
Transcribed Image Text:
Zn + Cu2+ Fe + 2H* Cl, + 2Br- Zn²+ + Cu Fe2+ + H2 2CI+ Br, 111
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (10 reviews)
Oxidized ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A disproportionation reaction is an oxidation-reduction reaction in which the same substance is oxidized and reduced. Complete and balance the following disproportionation reactions:
-
Complete and balance the following half-reactions. In each case indicate whether the half-reaction is an oxidation or a reduction.
-
Complete and balance the following half-reactions. In each case indicate whether the half-reaction is an oxidation or a reduction. Discuss.
-
You have been asked by the principal investigator of a qualitative research project to suggest ideas for maintaining confidentiality of private, sensitive information obtained from human...
-
Halle describes "Third-worldism" as an automatic resistance to change among poorer countries to proposals that come from richer countries. What could be done to overcome this distrust?
-
What role does the cost gap play in the attainment phase of target costing?
-
Refer to Exercise 5-4 and prepare the appropriate journal entries for DeSoto Co. to record the May 5 purchase and each of the three separate transactions a through c. DeSoto is a retailer that uses a...
-
The following interfund transactions occurred in Becca Citys General Fund during the year ended December 31, 2012. For each transaction, describe its nature (e. g., is it an interfund transfer, an...
-
Watch help video In circle.F with m/EFG = 70 and EF Round to the nearest hundredth. E F G 19 units, find the length of arc EG.
-
1. Find out consumer surplus and producer surplus of the Home country in autarky. (3pts) 2. Derive the import demand function of the Home country and the export supply function of the Foreign...
-
When magnesium is placed in an acid solution, hydrogen gas is given off. Is magnesium or hydrogen the better reducing agent?
-
Hydrogen sulfide gas dissolves in water and dissociates very slightly: H 2 S 2H + + S 2 -. How would the acidity of the solution be affected by a. Increasing the pressure of H 2 S? b. Raising the...
-
Construct a 95% prediction interval for an individual response when x = 20. Exercises 610 refer to the following data set: x 25 13 16 19 29 19 16 30 y 40 20 33 30 50 37 34 37
-
The National Practitioner Data Bank (NPDB) is used to collect and release information on the professional competence and conduct of physicians, dentists, and other healthcare practitioners....
-
Title VII of the Civil Rights Act of 1964, as amended by the Equal Employment Opportunity Act of 1972, prohibits discrimination in employment based on race, color, religion, sex, or national origin.
-
In 1974, an amendment designating procedures that limit strikes in healthcare organizations was added to the NLRA. The amendment requires a 10-day strike notice; allows the NLRB to determine the...
-
Fairly balancing the rights and responsibilities of the employee and the needs of the organization is an extremely complex objective. Rights and responsibilities run parallel to one another. There...
-
Staff responsibilities include compassion; honoring patient wishes; maintaining confidentiality for both patients and employees; adhering to safe practices (e.g., comply with sterile technique...
-
For (x) = 3x and g(x) = [1/4] x , find each of the following. Round answers to the nearest thousandth as needed. (-2)
-
4. Jobe dy -Y 2 et by
-
A vessel containing a liquid is opened inside an evacuated chamber. Will you see a liquidgas interface if the volume of the initially evacuated chamber is a. Less than the critical volume, b. A...
-
Calculate S R for the reaction H 2 (g) + Cl 2 (g) 2HCl (g) at 870. K. Omit terms in the temperature dependent heat capacities higher than T 2 /K 2 .
-
Use the result of Problem P3.10 to derive a formula for (CV /V ) T for a gas that obeys the RedlichKwong equation of state, RT 1 a Vm - b VT VVm + b)' T VVm P:
-
Following is information on two alternative investment projects being considered by Tiger Company. The company requires a 7% return from its investments. (PV of $1, FV of $1, PVA of $1, and FVA of...
-
5 23 Which of the following events would generally NOT require a company to record a current liability? Multiple Choice Assessment of Income Tax Expense at year-end. Sale of product to a customer,...
-
Farris Corporation, which has only one product, has provided the following data concerning its most recent month of operations: Selling price Units in beginning inventory Units produced Units sold...

Study smarter with the SolutionInn App


