A Nb-W alloy held at 2800C is partly liquid and partly solid. (a) If possible, determine the
Question:
A Nb-W alloy held at 2800°C is partly liquid and partly solid.
(a) If possible, determine the composition of each phase in the alloy, and
(b) If possible, determine the amount of each phase in the alloy.
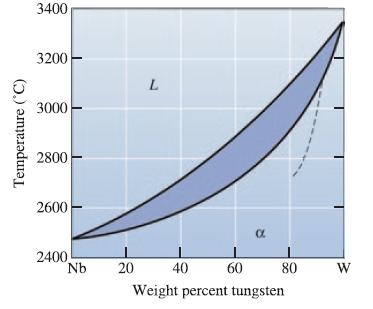
Transcribed Image Text:
Temperature (°C) 3400 3200 3000 2800 2600 2400 Nb L 20 α 40 60 Weight percent tungsten 80 W
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
a The composition of each phase in the al...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the eutectoid temperature, the composition of each phase in the eutectoid reaction, and the amount of each phase present in the eutectoid microconstituent for the following systems. For the...
-
Determine the composition of the products of the disassociation reaction CO2 CO + O when the products are at 1 atm and 2500 K.
-
A Nb-60 wt% W alloy is heated to 2800 C. Determine (a) The composition of the solid and liquid phases in both wt% and at%; (b) The amount of each phase in both wt% and at%; and (c) Assuming that the...
-
k) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. What is the highest utility score that can be achieved...
-
Starting from your graphical results of Problem 7b, show graphically the effect on YE and on XM of a depreciation of the nations currency from a position of full employment and a trade deficit.
-
PHB Company currently sells for $ 32.50 per share. In an attempt to determine whether PHB is fairly priced, an analyst has assembled the following information: The before - tax required rates of...
-
Five shareholders together own 35 percent of the outstanding stock of Edmonds Industries. The remaining 65% is divided among several thousand stockholders. There are 400,000 shares ol Edmonds stock...
-
Now reconsider the expanded version of the Silicon Dynamics problem described in Probs.16.3-2 and 16.4-2. (a) Use ASPE to construct and solve the decision tree for this problem. (b) Perform...
-
PARTA A Company produces and sells custom made Balls. The company uses a job order costing system and applies manufacturing overhead cost to jobs on the basis of direct labor hours. Its predetermined...
-
An FI has purchased a $200 million cap of 9 percent at a premium of 0.65 percent of face value. A $200 million floor of 4 percent is also available at a premium of 0.69 percent of face value. a. If...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in mol% for the following NiO-MgO ceramics at 2400C: (a) NiO- 30 mol% MgO; (b) NiO-45 mol% MgO; (c) NiO-60...
-
A Bi-60 at% Sb alloy is cooled under equilibrium conditions from the liquid state. Determine (a) The temperature at which the first solid nucleus forms and its composition; (b) The compositions and...
-
Employ the program from Prob. 29.12 to solve Probs. 29.1 and 29.2.
-
If Technical Specification 2 were reduced in the next design for this product, what would likely happen to customer opinion of Value Feature A? Quick Start QFD Matrix 2 Strong positive correlation...
-
Customer opinion of Value Feature B is most strongly correlated with what technical specification? Quick Start QFD Matrix 2 Strong positive correlation Some positive correlation == Strong negative...
-
Consider Quick Start QFD Matrix 1 above. Of the two value features, which do cus- tomers consider three times more important? Quick Start Quick Start QFD Matrix 1 = Strong positive correlation = Some...
-
Which technical spec can be most easily modified without changing current choices for the other two technical specs? Quick Start Quick Start QFD Matrix 1 = Strong positive correlation = Some positive...
-
Use Table A.1 to select 20 three-digit random numbers. Did any of the numbers occur more than once? How is it possible for a number to occur more than once? Make a stem-and-leaf plot of the numbers...
-
Write the vector v in the form ai + bj given its magnitude |v| and the angle it makes with the positive x-axis. |v| = 8, = 45
-
Tarick Toys Company manufactures video game consoles and accounts for product costs using process costing. The following information is available regarding its June inventories. The following...
-
The codons (words) in DNA (that specify which amino acid should be at a particular point in a protein) are three bases long. How many such three-letter words can be made from the four bases adenine,...
-
Part of a certain DNA sequence is GGTCTATAC. What is the complementary sequence?
-
Which of the noncyclic isomers of C 4 H 7 F are optically active?
-
nformation pertaining to Noskey Corporation s sales revenue follows: November 2 0 2 1 ( Actual ) December 2 0 2 1 ( Budgeted ) January 2 0 2 2 ( Budgeted ) Cash sales $ 1 0 5 , 0 0 0 $ 1 1 5 , 0 0 0...
-
The management team of Netflix maintains a stable dividend using the Lintner model: Dt+1 = Dt + EPS Target Payout Where Dt (Dt+1) = dividend in the current period t (the next period t + 1) EPSt =...
-
#1 #2 hapter 50 10 D Werences lav Help Required information [The following information applies to the questions displayed below) Archer Company is a wholesaler of custom-built air-conditioning units...

Study smarter with the SolutionInn App


