Consider the dissociation of a weak acid HA (K a = 4.5 10 -3 ) in
Question:
Consider the dissociation of a weak acid HA (Ka = 4.5 × 10-3) in water:
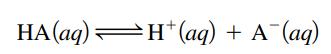
Calculate ΔGο for this reaction at 25οC.
Transcribed Image Text:
HA (aq) H+ (aq) + A¯(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
G for this reaction at 25C G for ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
A 200 Watt light bulb has a current of 0.7 A passing through it. The total charge that has passed through the light bulb after two hours is. Select the closest answer: O 1.4 C O 6734 C O 2520 C O...
-
Consider two separate aqueous solutions: one of a weak acid HA and one of HCl. Assuming you started with 10 molecules of each: a. Draw a picture of what each solution looks like at equilibrium. b....
-
A weak acid HA (pKa 5.00) was titrated with 1.00 M KOH. The acid solution had a volume of 100.0 mL and a molarity of 0.100 M. Find the pH at the following volumes of base added and make a graph of pH...
-
A local university is considering changes to its class structure in an effort to increase professor productivity. The old schedule had each professor teaching 5 classes per week, with each class...
-
Suppose oil prices jump up and the Fed is completely accommodative: it keeps the real interest rate constant. How must the Fed adjust the nominal interest rate? How must it adjust the money supply?...
-
Each of the following reactions has been reported in the chemical literature. Predict the product in each case, showing stereochemistry where appropriate. (a) CHs CH hea (b) (CH3)2(-C(CH3): T -...
-
Calculate \(3 \times 6^{3}-18\).
-
Kegglers Supply is a merchandiser of three different products. The companys February 28 inventories are footwear, 20,000 units; sports equipment, 80,000 units; and apparel, 50,000 units. Management...
-
Required information [The following information applies to the questions displayed below.] Built-Tight is preparing its master budget. Budgeted sales and cash payments follow: Budgeted sales July $...
-
(a) Consider an extended object whose different portions have different elevations. Assume the free-fall acceleration is uniform over the object. Prove that the gravitational potential energy of the...
-
For the equilibrium the initial concentrations are P A = P B = P C = 0.100 atm. Once equilibrium has been established, it is found that P C = 0.040 atm. What is G for this reaction at 25 C? A(g) +...
-
A copper penny can be dissolved in nitric acid but not in hydro chloric acid. Using reduction potentials from the book, show why this is so. What are the products of the reaction? Newer pennies...
-
What types of problems can occur when sections of a casting intersect?
-
Compare the process of budgeting revenues and expenses with the process of budgeting assets, liabilities, and owners equity, and explain how this information is included in the QuickBooks Accountant...
-
Describe the typical relationship between accumulated depreciation and depreciation expense and how this information is included in the QuickBooks Accountant budgeting process.
-
Explain how the Copy Across feature helps in creating QuickBooks Accountant budgets.
-
Explain how the Adjust Row Amounts feature helps in creating QuickBooks Accountant budgets.
-
Explain the typical relationship between sales and cost of goods sold, and describe how this information is included in the QuickBooks Accountant budgeting process.
-
In 2014, Laureen is currently single. She paid $2,800 of qualified tuition and related expenses for each of her twin daughters Sheri and Meri to attend State University as freshmen ($2,800 each for a...
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
Draw a segment of the polymer that might be prepared from 2-phenyl-1, 3-butadiene.
-
Show the mechanism of the acid-catalyzed polymerization of 1, 3-hutadiene.
-
Calculate the energy range of electromagnetic radiation in the UV region of the spectrum from 200 to 400 nm. How does this value compare with the values calculated previously for JR and NMR...
-
Cchegg G what is x G budge x How T X Sign o edu/d2l/le/content/107694/viewContent/5524273/View c) demand deposits d) money market mutual funds Question 50 (1 point) The Fed can influence unemployment...
-
Chapter 9 Assignment Questions 1. How many PowerShell profiles are available? 2. What is the $pshome variable? 3. Whats does running the variable $pshome in PS console produce? 4. What is the...
-
1. What is DSC? Chapter 21 Assignment Questions 2. What is a prerequisite for running DSC? 3. In PowerShell v5 how many types of native DSC resources are available 4. What is the file format used by...

Study smarter with the SolutionInn App


