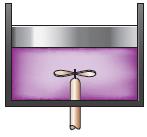
Question: A closed system like that shown in Fig. P427E is operated in an adiabatic manner. First, 15,000 lbfft of work are done by this system.
A closed system like that shown in Fig. P4–27E is operated in an adiabatic manner. First, 15,000 lbf·ft of work are done by this system. Then, work is applied to the stirring device to raise the internal energy of the fluid by 10.28 Btu. Calculate the net increase in the internal energy of this system.
Step by Step Solution
★★★★★
3.36 Rating (171 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

A pistoncylinder device involves expansion work and work input by a s... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


