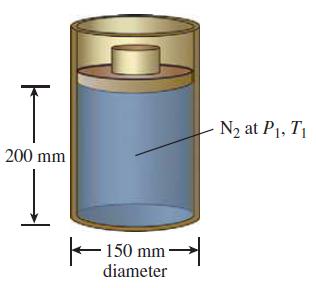
Consider nitrogen gas contained in a piston-cylinder arrangement as shown in the sketch. The piston is free
Question:
Consider nitrogen gas contained in a piston-cylinder arrangement as shown in the sketch. The piston is free to move without friction. The cylinder diameter is 150 mm. At the initial state the temperature of the N2 is 500 K, the pressure is 100 kPa, and the height of the gas column within the cylinder is 200mm. Heat is removed from the N2 by a quasi-static, constant-pressure process until the temperature of the N2 is 300 K. Calculate the work done by the N2 gas.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:





