Question:
Two adjacent tanks are connected by a valve. The tank on the left contains 0.5 kg of water at a pressure of 11 bar and a temperature of 600 K. The tank on the right contains 0.25 kg of saturated water at a temperature of 300 K and its quality is 0.4. If the valve between the tanks is opened and the tanks are allowed to reach thermal equilibrium with the environment, what is the amount of energy transfer as heat out of the system assuming the environment temperature is 22 ?C? Use the tables in Appendix A.3 for data.
Data Froxm Appendix A.3
Properties in tables and charts are calculated with IF97, which implements the thermodynamic model for water documented in W. Wagner and A. Kruse, Properties of Water and Steam: The Industrial Standard IAPWS-IF97 for the Thermodynamic Properties and Supplementary Equations for Other Properties. Springer-Verlag, 1998. Reference state: u0 = usat,L?? (Ttriple?= 273.16 K) = 0 kJ/kg, s0 = ssat,L??(Ttriple?= 273.16 K) = 0 kJ/ (kg ? K).
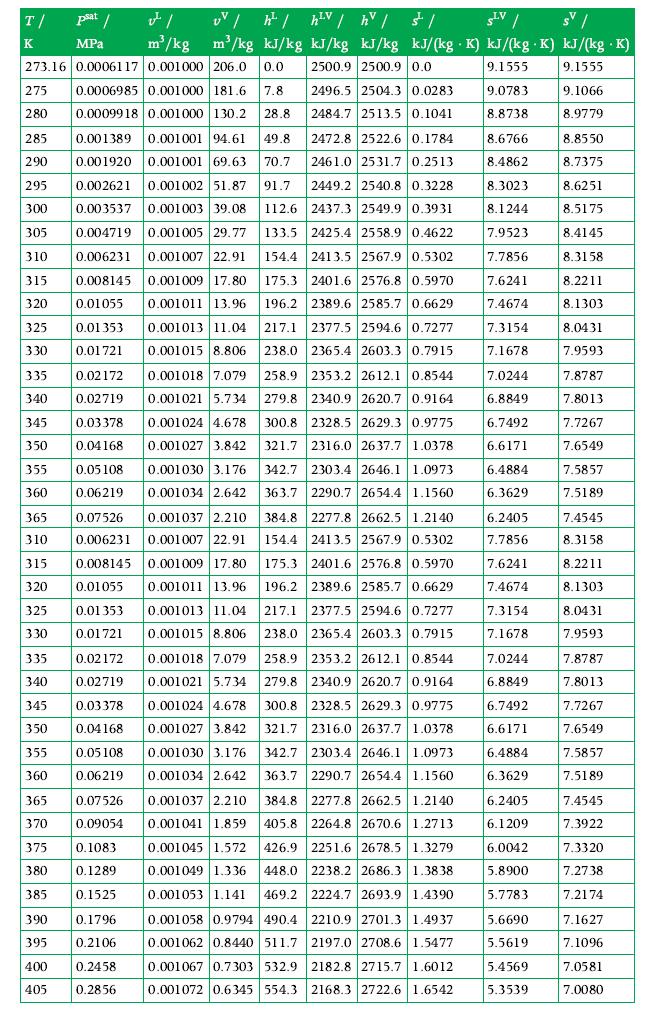
Transcribed Image Text:
T/ psat /
MPa
K
v
m³/kg
273.16 0.0006117 0.001000 206.0 0.0
0.0006985
0.001000 181.6 7.8
0.0009918 0.001000 130.2 28.8
335
340
345
350
355
360
UV /h²/h²V / hv /
/
SLV /
sv/
m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg K) kJ/(kg.K) kJ/(kg .K)
2500.9 2500.9 0.0
9.1555
9.1555
9.0783
8.8738
275
280
285
0.001389 0.001001 94.61 49.8
290
0.001920
0.001001 69.63 70.7
295 0.002621 0.001002 51.87 91.7
300
0.003537
0.001003 39.08 112.6
305
0.004719
0.001005 29.77 133.5
310
0.006231
0.001007 22.91
0.001009 17.80 175.3
315 0.008145
320 0.01055
0.001011 13.96
325
0.01 353
0.001013 11.04
330
0.01721
0.001015 8.806
0.02172
0.02719
0.03378
0.04168
0.05108
0.06219
0.07526
0.006231
365
310
325
330
0.001018 7.079
0.001021 5.734
0.001024 4.678
0.001027 3.842
0.001030 3.176
0.001034 2.642
0.001037 2.210
0.001007 22.91
315 0.008145 0.001009 17.80
320
0.01055
0.001011 13.96
0.01 353
0.001013 11.04
0.01721
0.001015 8.806
335 0.02172
0.001018 7.079
0.001021 5.734
340 0.02719
345 0.03378
350 0.04168
355 0.05108
360 0.06219
365 0.07526
0.001024 4.678
0.001027 3.842
0.001030 3.176
0.001034 2.642
370 0.09054
375 0.1083
380 0.1289
385 0.1525
390 0.1796
395 0.2106
400 0.2458
405 0.2856
2496.5 2504.3 0.0283
2484.7 2513.5 0.1041
2472.8 2522.6 0.1784
2461.0 2531.7 0.2513
2449.2 2540.8 0.3228
2437.3 2549.9 0.3931
2425.4 2558.9 0.4622
154.4 2413.5 2567.9 0.5302
2401.6 2576.8 0.5970
196.2 2389.6 2585.7 0.6629
217.1 2377.5 2594.6 0.7277
238.0 2365.4 2603.3 0.7915
258.9 2353.2 2612.1 0.8544
279.8 2340.9 2620.7 0.9164
2328.5 2629.3 0.9775
300.8
321.7
2316.0 2637.7 1.0378
2303.4 2646.1 1.0973
342.7
363.7 2290.7 2654.4 1.1560
2277.8 2662.5 1.2140
384.8
154.4 2413.5 2567.9 0.5302
175.3
2401.6 2576.8 0.5970
196.2
2389.6 2585.7 0.6629
217.1
2377.5 2594.6 0.7277
238.0
2365.4 2603.3 0.7915
258.9
2353.2 2612.1 0.8544
2340.9 2620.7 0.9164
279.8
300.8 2328.5 2629.3 0.9775
321.7
342.7 2303.4 2646.1 1.0973
363.7 2290.7 2654.4 1.1560
2316.0 2637.7 1.0378
384.8 2277.8 2662.5 1.2140
405.8 2264.8 2670.6 1.2713
0.001037 2.210
0.001041 1.859
0.001045 1.572 426.9 2251.6 2678.5 1.3279
0.001049 1.336
0.001053 1.141 469.2
0.001058 0.9794 490.4 2210.9 2701.3 1.4937
0.001062 0.8440 511.7 2197.0 2708.6 1.5477
0.001067 0.7303 532.9 2182.8 2715.7 1.6012
0.001072 0.6345 554.3 2168.3 2722.6 1.6542
448.0 2238.2 2686.3 1.3838
2224.7 2693.9 1.4390
8.6766
8.4862
8.3023
8.1244
7.9523
7.7856
7.6241
7.4674
7.3154
7.1678
7.0244
6.8849
6.7492
6.6171
6.4884
6.3629
6.2405
7.7856
7.6241
7.4674
7.3154
7.1678
7.0244
6.8849
6.7492
6.6171
6.4884
6.3629
6.2405
6.1209
6.0042
5.8900
5.7783
5.6690
5.5619
5.4569
5.3539
9.1066
8.9779
8.8550
8.7375
8.6251
8.5175
8.4145
8.3158
8.2211
8.1303
8.0431
7.9593
7.8787
7.8013
7.7267
7.6549
7.5857
7.5189
7.4545
8.3158
8.2211
8.1303
8.0431
7.9593
7.8787
7.8013
7.7267
7.6549
7.5857
7.5189
7.4545
7.3922
7.3320
7.2738
7.2174
7.1627
7.1096
7.0581
7.0080







