The exit gas from an alcohol fermenter consists of an air-C02 mixture containing 10 mol% C02 that
Question:
The exit gas from an alcohol fermenter consists of an air-C02 mixture containing 10 mol% C02 that is to be absorbed in a 5.0-N solution of triethanolamine, containing 0.04 mol of carbon dioxide per mole of amine solution. If the column operates isothermally at 25oC, if the exit liquid contains 78.4% of the C02 in the feed gas to the absorber, and if absorption is carried out in a six-theoretical-plate column, calculate:(a) Moles of amine solution required per mole of feed gas.(b) Exit gas composition.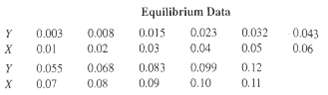 Y = moles C02/mole air; X = moles C02/mole aminesolution
Y = moles C02/mole air; X = moles C02/mole aminesolution
Transcribed Image Text:
Equilibrium Data 0.032 0.05 0.12 0.008 0.023 0.003 0.043 0.015 0.04 0.06 0.02 0.03 0.083 0.01 0.055 0.07 0.068 0.099 0.10 х 0.09 0.11 0.08
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (21 reviews)
Use the nomenclature and type of plot shown in Fig a Therefore for CO 2 b There...View the full answer

Answered By

Atuga Nichasius
I am a Highly skilled Online Tutor has a Bachelor’s Degree in Engineering as well as seven years of experience tutoring students in high school, bachelors and post graduate levels. I have a solid understanding of all learning styles as well as using asynchronous online platforms for tutoring needs. I individualise tutoring for students according to content tutoring needs assessments.
My strengths include good understanding of all teaching methods and learning styles and I am able to convey material to students in an easy to understand manner. I can also assists students with homework questions and test preparation strategies and I am able to help students in math, gre, business , and statistics
I consider myself to have excellent interpersonal and assessment skills with strong teaching presentation verbal and written communication
I love tutoring. I love doing it. I find it intrinsically satisfying to see the light come on in a student's eyes.
My first math lesson that I taught was when I was 5. My neighbor, still in diapers, kept skipping 4 when counting from 1 to 10. I worked with him until he could get all 10 numbers in a row, and match them up with his fingers.
My students drastically improve under my tutelage, generally seeing a two grade level improvement (F to C, C to A, for example), and all of them get a much clearer understanding!
I am committed to helping my students get the top grades no matter the cost. I will take extra hours with you, repeat myself a thousand times if I have to and guide you to the best of my ability until you understand the concept that I'm teaching you.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Exit gas from an amination reactor contains 10 mole% ammonia (NH 3 ) vapor in a nitrogen (N 2 ) carrier gas. This gas mixture is fed into the bottom of a packed tower at a molar flow rate of 2.0...
-
The back of a framed picture that is to be hung is shown at the top of the next page. A nail is to be hammered into the wall, and the picture will be hung by the wire on the nail. a) If the center of...
-
A program file contains data that is to be used by various programs. True of False
-
Give examples of three exceptions to the Cardozo rule of foreseeability.
-
The unadjusted trial balance of Fashion Centre Ltd. contained the following accounts at November 30, the company's fiscal year end: Additional information and adjustment data: 1. The 12-month...
-
Western Electric Company pays its support staff weekly and its electricians on a semi-monthly basis. The following support staff payroll information is available for the week ended June 9, 2018: The...
-
How can software assist in project stakeholder management? Do you think social media tools are more likely to help or hinder projects? LO.1
-
Wayne Kaegi's Verde Vineyards in Oakville, California, produces three varieties of wine: Merlot, Viognier, and Pinot Noir. His winemaster, Russel Hansen, has identified the following activities as...
-
Auditing Jiang Chao, CPA, is the auditor of Ellice Pearls. Jiang is considering the audit work to be performed in the accounts payable area for the current year engagement. After obtaining an...
-
Paste Clipboard A1 3 4 5 6 1.000 7 8 9 10 11 FILE 2 12 13 14 15 5 6 16 1. Prepare a bank reconciliation using a company's bank statement and cash account. HOME Date June 1 3 8545NOWO A 10 13 A B ...
-
Prove by equations why, in general, absorbers should be operated at high pressure and low temperature, while strippers should be operated at low pressure and high temperature. Also prove, by...
-
Ninety-five percent of the acetone vapor in an 85 vol% air stream is to be absorbed by countercurrent contact with pure water in a valve-tray column with an expected overall tray efficiency of 50%....
-
To four decimal places, the values of log 10 2 and log 10 9 are Use these values and the properties of logarithms to evaluate each expression. log10 2 = 0.3010 and log10 9 = 0.9542.
-
Medical Helicopters In a study of helicopter usage and patient survival, results were obtained from 47,637 patients transported by helicopter and 111,874 patients transported by ground (based on data...
-
On January 1, 20X1, Laketown Company (the user) leased a truck for a seven-year period under a FINANCE LEASE and agreed to pay an annual lease payment of $6,000 at the end of each year. The interest...
-
ces Shouldice Hospital in Canada is widely known for one thing-hernia repair! In fact, that is the only operation it performs, and it performs a great many of them. Over the past two decades this...
-
2. An Annual General Meeting (AGM) is a meeting conducted annually where the members of an organization gather to discuss and vote on key issues. Public companies hold annual general meetings for...
-
The 3 P's of Sustainability refers to: O The groups of people that environmental efforts effect. O People, Planet, Profit O Population, Productivity, Principled None of the Above. Question 21 2...
-
What are the various styles used when setting up a meeting room? Give examples of when each style might be used.
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
Why do scholars suggest that we are unlikely to see a convergence of HRM practices to one best model?
-
In a filtration cycle, why does constant-pressure filtration usually occur near the end of the cycle and constant-rate filtration at the beginning?
-
A cyclone of diameter D c of 2 ft, whose dimension ratios are as given in Figure 19.9, is being considered to remove dust from a cement kiln. The gas feed, at inlet velocity i of 20 ft 3 /s, has 0.5...
-
Why have theoretical analyses that treat voids in filter cakes as flow channels not been applied industrially?
-
For esch of the following Independent tranactiona, determine the minimum amount of net income or loas for tox purposes snd the tsxpsyer to which it applies. 1 An individual purchases a $ 1 0 , 0 0 0...
-
Suppose a bond has a modified duration of 4. By approximately how much will the bonds value change if interest rates: a. Increase by 50 basis points b. Decrease by 150 basis points c. Increase by 10...
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...

Study smarter with the SolutionInn App


