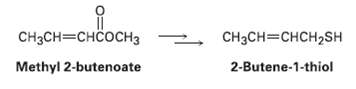
2-Butene-1-thiol is one component of shunk spray. How would you synthesize this substance from methyl 2-butenoate? From
Question:
2-Butene-1-thiol is one component of shunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene
Transcribed Image Text:
CH3CH=CHCOCH3 CH3CH=CHCH2SH 2-Butene-1-thiol Methyl 2-butenoate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
Thiourea is used to prepare thiols from alk...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you synthesize the following esters? a. N-octylacerate b. a404040404040-20H CH,CH2CH2CH2CH2CH2O-CCH2CH3
-
How would you synthesize the following compounds from Cyclohexanone? More than one step may berequired. () CH2 CH2Br (a) (c) o CH2C6H5 CH2CH2CO2H (d) (e) (f) O
-
How would you synthesize the following alcohols, starting with benzene and other alcohols of six or fewer carbons as your only organic reagents? CH3 (a) (b) CH3CH2CH2HCH2CH2OH "CH-CH (c) (d) CH ...
-
Translate the seven steps to Java code.
-
The checkpoints U.S. citizens pass through upon returning to the country vary greatly in the depth of the checks and the time spent. The simplest involves simply walking past the border guards who...
-
Gfeller Enterprises began fiscal 2010 with the following shareholders equity information: Common stock shares issued 39,598,900, $ 0.01 par value, paid-in capital in excess of par value, $...
-
Determine whether the following are preventive (P), detective (D), or corrective (C) controls. a. Training b. Bank reconciliations c. Passwords d. Surprise cash count e. Quality circle team f. Job...
-
The following selected transactions were complete by Lawn supplies co., which sells irrigation supplies primarily to wholesalers and occasionally to retail customers: Mar. 1. Sold merchandise on...
-
PROBLEM 1 A company sells two products, Product 1 and Product 2. Three units of Product 1 are sold for every two units of Product 2. Fixed costs is P234,000 per year. Product 1 is sold P20 per unit...
-
William Gail opened his automobile cleaning service G-Force Auto Detailing, specializing in interior cleaning of automobiles. The end-of-year unadjusted trial balance contained the following accounts...
-
Name the followingcompounds: CH (b) (c) SH (a) CH SH CH CH3CH2CHSH CHH-CHH2H CH (e) C (d) (f) SCH3 CHCHSCH2CH3 SCH3 SCH2CH3
-
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
-
Hansen Industries reported the following results for 20x6. REQUIRED: Calculate the industry volume variance and market share variance for the year. Hansen Industries Budget Actual Industry Volume...
-
1. Describe each of the four stages in Piaget's theory. Give examples of the following and indicate the stage when each occurs: object permanence, egocentrism, conservation, abstract reasoning. What...
-
An open flat belt is to transmit 8 kW. The small pulley has a diameter of 175 mm and rotates at 810 r/min, whilst the large pulley runs at approximately 250 r/min. Both pulleys have a width of 110...
-
1. What information does the Institute for Safe Medication Practices provide to help nurses minimize the risk of medication errors? Give three specific examples. Provide a rationale to support your...
-
1. Discuss how a consensus model and a conflict model of criminal justice develop in a given society (e.g. U.S.). Give examples, while discussing the pros and cons of each. Discuss the advantages and...
-
Define promotion and integrated marketing communication (IMC). Discuss a basic model of communication.. Describe the different ways of classifying audiences for promotion and IMC. Explain the key...
-
What acts are considered discreditable to the profession under Rule 501?
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
A sugar mill in Florida has been disposing of the bagasse (used sugar cane) by open-air burning. You, as a new chemical engineer, determine that by using the dried bagasse as boiler fuel in the mill,...
-
Explain how different signals may would appear in the 13 C-NMR- spectra of these compounds.
-
Anthracene has 1.80 x 105 M1 cm1 at max = 256nm calculate the absorbance of a 1.94 x 106M solution of anthracene in a 1 cm cell.
-
A solution of a 0.0014 g of benzophenone in 1 L of ethanol has A = 0.153 (1 cm cell) at ? max = 252 nm. Calculate the molar absorptivity of benzophenone. Benzophenone
-
Banks earn profits by selling ____ using the proceeds to buy securities; deposits assets; liabilities liabilities; assets loans; deposits with attractive combinations of liquidity, risk, and return,...
-
The following information pertains to The Unique Boutique Company. Assume that all balance sheet amounts represent both average and ending balance figures. Assume that all sales were on credit. image...
-
When constructing a pro forma statement, net working capital generally: A. remains fixed. B. varies only if the firm is currently producing at full capacity. C. varies only if the firm maintains a...

Study smarter with the SolutionInn App


