A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes
Question:
A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:
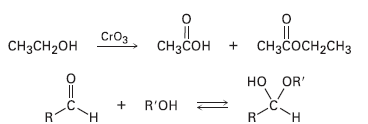
Transcribed Image Text:
|| CHзCон + снзҫосн-сHз CrOз CH3CH2OH но OR' + R'OH .C. н н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
An alcohol adds to an aldehyde by a mechanism that we will study in ...View the full answer

Answered By

Joan Gakii
I'm a meticulous professional writer with over five years writing experience. My skill set includes
- Digital Content,
- Interpersonal Communication,
- Web Content and academic Writing,
- Proofreading,
- Editing,
- Project Management, and
- Public Relations.
5.00+
7+ Reviews
12+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
Propose a mechanism to account for the formation of Bakelite from acid-catalyzed polymerization of phenol and formaldehyde.
-
Overoxidation of primary alcohols to carboxylic acids is caused by the water present in the usual aqueous acidic Cr(VI) reagents. The water adds to the initial aldehyde product to form a hydrate,...
-
Bollobs and Chung proposed a hybrid model that combines a 2-ring on \(V\) vertices ( \(V\) is even), plus a random matching. A matching is a graph in which every vertex has degree 1. To generate a...
-
Which of the three basic philosophies of social responsibility would you find most appealing as the chief executive of a large corporation? Explain.
-
Does the Retained Earnings account contain cash? Explain your answer.
-
What three certifications does the National Association of Certified Valuation Analysts offer?
-
The Pitts Barbecue Company makes three kinds of barbecue sauce: Extra Hot, Hot, and Mild. Pitts vice president of marketing estimates that the company can sell 8,000 cases of its Extra Hot sauce plus...
-
............... The following information is extracted from annual reports of Sunshine Lid at the end of 2020. 2020 2019 $ $ Cash assets 22,000 30,000 Accounts Receivable 33,000 40,000 Inventory...
-
1. The university rules allow an F grade to be overridden by any pass grade (A, B, C, D). Now, create a view that lists information about all fail grades that have not been overridden (the view...
-
Propose a synthesis of bicyclohexylidene, starting from Cyclohexanone as the only source of carbon. Bicyclohexylidene
-
Identify the reagents a?f in the following scheme: .Br CH-
-
The Gold Company is applying for a five-year term loan from its bank. The lender determines that the firm should pay a default risk premium of 1.75 percent over the Treasury rate. The five-year...
-
Q3. (a) Prove that Sin(A+B)Sin(A-B)=sin A-sin B = cos B-cos A (b) Find x from the equation: cosec(90 + A+xcos A cot(90 +4)=sin(90 + 4)
-
Rainbow Salon had the following data for the most recent three months of operations. What is Rainbow's cost equation? May June July of appointments 1,200 1,000 1,400 Hair dresser salaries $30,000...
-
The U.S. economy is starting to find its way through the economic turmoil caused by the pandemic. Government policy was very active with both monetary policy and fiscal policy attempting to keep the...
-
The case study is a Mass Casulty incident fire and the role is Patient Experience Manager at a Hospital Lessons Learned Report Directions. Your Report should address each Aspect requirement as...
-
7. Name two types of errors that might occur in the inventory account. If the error understates beginning inventory, how is cost of good sold impacted? If the error understates ending inventory, how...
-
LM produces two products from different quantities of the same resources using a just-in-time (JIT) production system. The selling price and resource requirements of each of these two products are as...
-
Kims Konstructions has assembled the following data for a proposed straw-reinforced brick maker (SRBM): SRBM Cost: $26,000 Life: 5 years Revenue (p.a.) $11,000 Operating Expenses (p.a.) $3,000...
-
Is there any scientific basis for the colloquial expression slower than molasses in January? Explain.
-
When (S)-2-bromopropanoic acid [(S)-CH3CHBrCO2H] reacts with concentrated sodium hydroxide, the product formed (after acidification) is (R)-2-hydroxypropanoic acid [(R)-CH3CHOHCO2H, commonly known as...
-
Using chair conformational structures (Section 4.11), show the nucleophilic substitution reaction that would take place when trans-1-bromo-4-tert-butylcyclohexane reacts with iodide ion. (Show the...
-
The phenomenon of configuration inversion in a chemical reaction was discovered in 1896 by Paul Walden (Section 6.6). Walden's proof of configuration inversion was based on the following cycle: (a)...
-
What ingredients are needed to power a dynamo effect inside a planet? Group of answer choices A liquid iron core with a high density that is covered by a thick layer of liquid metallic hydrogen. A...
-
Physical mutagens include ______. Multiple choice question. neither X-rays nor UV light both X-rays and UV light UV light but not X-rays X-rays but not UV light
-
True or False: Percentage body fat can be measured by using calipers, bioelectrical impedance, dual-energy X-ray absorptiometry, underwater weighing, or air displacement. Ask Studybuddy Group of...

Study smarter with the SolutionInn App


