Identify the reagents a?f in the following scheme: .Br CH-
Question:
Identify the reagents a?f in the following scheme:
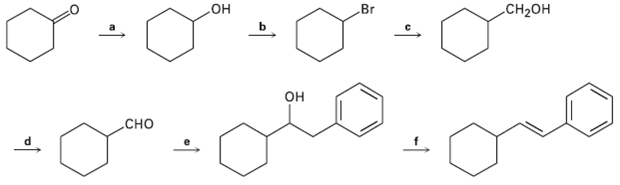
Transcribed Image Text:
.Br CH-Он но он Сно
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
CHOH d OH b CHO Br OH a ...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify reagents that can be used to achieve each of the following transformations: a. b. c. d. e. f. Br Br HO,
-
Identify reagents that can be used to achieve each of the following transformations: a. b. c. Br
-
Identify reagents necessary to convert cyclohexane into 1,3-cylohexadiene: Notice that the starting material has no leaving groups and cannot simply be treated with a strong base. You must first...
-
Write a BST client that uses the data file ip-tocountry.csv found on the booksite to determine the source country of a given IP address. The data file has five fields: beginning of IP address range,...
-
Do you think a business organization in today's society benefits by defining a socially responsible role for itself? Why or why not?
-
Bradfield Manufacturing is considering three capital investment proposals. At this time, Bradfield Manufacturing has funds available to pursue only one of the three investments. Requirement Which...
-
ABC, Inc. has a target debt-equity ratio of 0.4. Its cost of equity is 16 percent and its cost of debt is 8 percent. If the tax rate is 32 percent, what is ABC's WACC? a. 10.3847% b. 11.6723% c....
-
Keynote Inc. uses a calendar year for financial reporting. The company is authorized to issue 20,000,000 shares of $1 par common stock . At no time has Keynote issued any potentially dilutive...
-
Lavage Rapide is a Canadian company that owns and operates a large automatic car wash facility near Montreal. The following table provides estimates concerning the company's costs: Fixed Cost per...
-
Rodeo & Blue Inc (RBI) has been a remarkable success story over the past four years. Starting from a small basement operation, it has quickly grown to a worldwide leader in the music industry. It is...
-
A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate:...
-
Galactose, a constituent of the disaccharide lactose found in dairy products, is metabolized by a pathway that includes the isomerization of UDP-galactose to UDP-glucose, where UDP = uridylyl...
-
Solve Prob. 17.10 assuming that the initial angular velocity of the flywheel is 360 rpm clockwise. Problem 17.10: The 200-mm-radius brake drum is attached to a larger flywheel that is not shown. The...
-
Write an essay why your doctor should care about social Justice ( In the United States) Healthcare should be the focus with concrete example Please use the following sources in the essay: 1). Artiga,...
-
Use the following references to formulate your answer to the question below: American Hospital Association. (2012). Managing population health: The role of the hospital . Retrieved from...
-
Using Digital Tools to Enhance Your Job Search Just as electronic media have changed the way candidates seek jobs, these same digital tools are transforming the way employers select qualified...
-
Gourmet Juices is a company that sells a variety of natural fruit juices. The company has enjoyed record-breaking profits; its products have earned several local and international awards. However,...
-
Determine fundamental approaches to scientific research in addressing questions related to the natural world Develop questions about fundamental aspects of the natural world that inform personal...
-
What are three examples of business model innovation using robots?
-
QUESTION 9 HC-O-C-R R-C-O-CH HC-O-P-O-CH-CH-NH3* O || O a. Phosphatidic acid, Serine O b. Lysophosphatidic acid, Serine, Free FA O c. Lysophosphatidylserine, Free FA O d. 2 Free FAs, Serine, Glycerol...
-
(A) The enthalpy of sublimation of cesium is 78.2 kJ mol -1 , and f H[CsCl(s)] = -442.8 kJ mol -1 . Use these values, together with other data from the text, to calculate the lattice energy of...
-
(R)-(3-Chloro-2-methylpropyl) methyl ether (A) on reaction with azide ion (N3-) in aqueous ethanol gives (S)-(3-azido-2-methylpropyl) methyl ether (B). Compound A has the structure...
-
Predict the structure of the product of this reaction: The product has no infrared absorption in the 1620-1680-cm-1 region. NaOH in aqueous EtOH CI HS
-
Cis-4-Bromocyclohexanol racemic C6H10O (compound C) Compound C has infrared absorption in the 1620-1680-cm-1 and in the 3590-3650-cm-1 regions. Draw and label the (R) and (S) enantiomers of product C.
-
In job-order costing system Question 1 oWhat is correct about job-order system? Question 2 options: all day to day expenses related to manufacturing of product by the firm are treated as factory...
-
What is the main purpose of a photographer?Select one:a.To write a script for the show.b.To finance and handle the production and cost needed in order to publish a print material.c.To capture images...
-
Accounting I15) SO ICEY Inc. has a periodic inventory system. The company purchased 225 units ofinventory at $11.50 per unit and 350 units at $12.50 per unit. What is the weightedaverage unit cost...

Study smarter with the SolutionInn App


