Although this biphenyl is chiral, it cannot be resolved. Explain. HO-C CH30 CO H OCH
Question:
Although this biphenyl is chiral, it cannot be resolved. Explain.
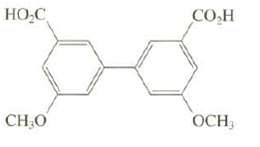
Transcribed Image Text:
HO-C CH30 CO H OCH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
The compound does not have a symmetry plane so it ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Biphenyl has the following structure. (a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon? (b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number...
-
Biphenyl is two benzene rings joined by a single bond. The site of substitution for a biphenyl is determined by (1) which phenyl ring is more activated (or less deactivated), and (2) which position...
-
It is possible for a compound to be chiral even though it lacks a carbon atom with four different groups. For example, consider the structure of the following compound which belongs to a class of...
-
Majan International School, Muscat conducts online classes and online exams for its students due to Covid 19 pandemic situation. It uses its own online Learning Teaching Management system namely...
-
Job costing: consulting firm WA Consulting Services Pty Ltd (WACS) provides consulting services throughout Western Austral and uses a job costing system to accumulate the cost of client projects....
-
In the Managerial Solution, would it make a difference to the analysis whether the lump-sum costs such as registration fees are collected annually or only once when the firm starts operation? How...
-
Explain why a companys manager wants the accounting system to record customers returns of unsatisfactory goods in the Sales Returns and Allowances account instead of the Sales account. In addition,...
-
Determine the magnitude of force F so that the resultant FR of the three forces is as small as possible. What is the minimum magnitude of FR? Units Used: kN = 1000 N Given: F1 = 5 kN F2 = 4 kN...
-
a) 1, 7, 8, 9, 11, 13, 15, 16, 22 b) 4, 5, 6, 6, 6, 7, 7, 8 c) -1.1, -1.1, -0.8, -0.5, -0.2, 0.1, 0.2, 0.5, 0.5 2) Using the student's t-distribution and assuming a 95% confidence interval, what is...
-
Suppose comm sz = 8 and n = 16. a. Draw a diagram that shows how MPI Scatter can be implemented using tree-structured communication with comm sz processes when process 0 needs to distribute an array...
-
Explain whether each of these compounds is chiral or not: a) d) H C H c=c=c CHCH CCH HC PhwSi-OCH-CH b) CHCH Ph-N-CHCH, HC Br H C CH30 CH CO H c) f) H H HC CH30 CCH CH3 COH CH3 CO H
-
Assign the configuration of these compounds as R or S: a) c) e) CH3 HC C HOCH H HC g) H- B CH CH3 i) HN- -OH CH H CH3 COH FH H CH,Ph Phenylalanine b) d) f) Br CH3 "CO,H h) H- me H to Br OH -CH3...
-
A wave pulse travels along a horizontal string. As the pulse passes a point on the string, the point moves vertically up and then back down again. How does the vertical speed of the point compare to...
-
Question 25 (6 points) Susan's Silken Scarves Ltd. produces scarves in a variety of designer colours. 2021 Master budget data is as follows: Quarter Sales (all on account) Raw material purchases 1 2...
-
A bank has had average profits of $35,675,000 over three years. Based on the BIA approach, what would the operational risk charge be?
-
A bank has multiple lines of business, with a three-year average net income of $58,750.000, categorized as follows: retail banking, 45%; corporate finance, 5%; agency (trust) services, 15%; brokerage...
-
1.Pacific Wind and Solar pays a dividend at the end of the year of $6 per share, but over the year, its stock price falls from $100 to $98. What is the annual rate of return for a shareholder in the...
-
You will have $180,000 saved in your retirement fund when you retire. If you put the savings into a payout annuity offering 6.5% interest and make monthly withdrawals for 20 years , what will your...
-
Forward error correcting codes are most likely to be used to detect line errors with which of the following transmission media? a. Coaxial cable b. Infrared c. Optical fiber d. Satellite microwave
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
In Exercises 38, compute (a) in two ways, using Eq. (1) and Eq. (2). (x) = x 2 + 9x, a = 2 f'(a) = lim h0 f(a+h)-f(a) h
-
Suppose that acetic acid (CH3CO2H) reacts with (S)-2-butanol to form an ester (see Worked Example 9.6). What stereochemistry would you expect the product(s) to have? What is the relationship of...
-
What stereoisomers would result from reaction of ()-lactic acid with (S)-1-phenyl- ethylamine, and what is the relationship between them?
-
What kinds of isomers are the following pairs? (a) (S)-5-Chloro-2-hexene and Chiorocyclohexane (b) (2R, 3R)-Dibromopentane and (2S, 3R)-dibromopentane
-
Accounting I2) During the first year of operations, BOOTIE LLC. Earned a net income totaling$30,400 and paid ordinary dividends of $12,200. In year 2, they reported net income of$34,400, paid...
-
For the following transactions/events , where relevant: a) Show all your calculations. b) Show all relevant journal entries c) Identify, provide and discuss the applicable accounting standard(s) for...
-
If you sell a product for $100, and your cost for producing the product is $30, what is your gross margin on that product? 30 percent gross margin 50 percent gross margin 70 percent gross margin None...

Study smarter with the SolutionInn App


