It is possible for a compound to be chiral even though it lacks a carbon atom with
Question:
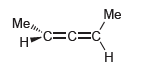
Transcribed Image Text:
Me Мет. HC=c=C Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
The following two compounds are enantiomers becau...View the full answer

Answered By

Saikumar Ramagiri
Financial accounting:- Journal and ledgers, preparation of trail balance and adjusted trail balance Preparation of income statement, retained earning statement and balance sheet Banks reconciliation statements Financial statement analysis Cash flow statement analysis (both direct and indirect methods) All methods of Depreciations Management Accounting:- Ratios Budgeting control Cash budget and production budget Working capital management Receivable management Costing:- Standard and variance costing Marginal costing and decision making Cost-volume-profit analysis Inventory management (LIFO, FIFO) Preparation and estimation of cost sheet Portfolio management:- Calculation of portfolio standard deviation or risk Calculation of portfolio expected returns CAPM, Beta Financial management:- Time value of money Capital budgeting Cost of capital Leverage analysis and capital structure policies Dividend policy Bond value calculations like YTM, current yield etc International finance:- Derivatives Futures and options Swaps and forwards Business problems Finance problems Education (mention all your degrees, year awarded, Institute/University, field(s) of major): Education Qualification Board/Institution/ University Month/Year of Passing % Secured OPTIONALS/ Major ICWAI(inter) ICWAI inter Pursuing Pursuing - M.com(Finance) Osmania University June 2007 65 Finance & Taxation M B A (Finance) Osmania University Dec 2004 66 Finance & Marketing. B.Com Osmania University June 2002 72 Income Tax, Cost & Mgt, Accountancy, Auditing. Intermediate (XII) Board of Intermediate May 1999 58 Mathematics, Accountancy, Economics. S S C (X) S S C Board. May 1997 74 Mathematics, Social Studies, Science. Tutoring experience: • 10 year experience in online trouble shooting problems related to finance/accountancy. • Since 6 Years working with solution inn as a tutor, I have solved thousands of questions, quick and accuracy Skills (optional): Technical Exposure: MS Office, SQL, Tally, Wings, Focus, Programming with C Financial : Portfolio/Financial Management, Ratio Analysis, Capital Budgeting Stock Valuation & Dividend Policy, Bond Valuations Individual Skills : Proactive Nature, Self Motivative, Clear thought process, Quick problem solving skills, flexible to complex situations. Achievements : 1. I have received an Award certificate from Local Area MLA for the cause of getting 100% marks in Accountancy during my Graduation. 2. I have received a GOLD MEDAL/Scholarship from Home Minister in my MBA for being the “Top Rank student “ of management institute. 3. I received numerous complements and extra pay from various students for trouble shooting their online problems. Other interests/Hobbies (optional): ? Web Surfing ? Sports ? Watching Comics, News channels ? Miniature Collection ? Exploring hidden facts ? Solving riddles and puzzles
4.80+
391+ Reviews
552+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structure of the following compound: (a) When this compound is treated with bromine under conditions that favor monobromination, two stereoisomeric products are obtained. Draw them, and...
-
Consider the structure of the following cyclic octapeptide. Would cleavage of this peptide with trypsin produce different fragments than cleavage with chymotrypsin? Explain. Phe Arg Arg Phe Phe Arg...
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
In terms of Federal tax consequences, why are lifetime transfers to charity preferable to testamentary transfers?
-
Here is the textbook by Daft, Richard, "Organization Theory and Design" 12th Cengage Learning, 2016. Chapter 10 discusses the components that create an organizational culture along with the various...
-
A newly incorporated town incurred and paid compensated absences for vacation pay and sick leave of $40,000 during its first year of operation, 20X1. 1. At the end of 20X1, the town estimated that...
-
The trial balance for BFF Systems, Inc., at March 15, 2010, follows: During the remainder of March, BFF Systems, Inc., completed the following transactions: Requirements 1. Journalize the...
-
The Ashland Company recently hired you to review its control procedures for the purchase, receipt, storage, and issuance of raw materials. You prepared the following comments, which describe Ashlands...
-
Math budget and tax #3 Assume the federal standard deduction is $6,300 and the federal personal exemption is $4,050. Single Taxable Income Brackets and Rates, 2017 Rate 10% Taxable Income Bracket 15%...
-
Wayland Custom Woodworking is a firm that manufactures custom cabinets and woodwork for business and residential customers. Students will have the opportunity to establish payroll records and to...
-
For each of the following pairs of compounds, determine the relationship between the two compounds. a. b. Et Me Me
-
Based on your analysis in the previous problem, determine whether the following allene is expected to be chiral: Me , Me, c=c=c Me
-
An increase in a persons net worth plus living expenses equals the persons total income. (True/False)
-
A centrifugal pump with an 8 in. diameter impeller operating at a rotational speed of $1150 \mathrm{rpm}$ requires $1.5 \mathrm{hp}$ to deliver water at a rate of $100 \mathrm{gpm}$ and a pressure of...
-
As a marketer, if you can find ways to classify your company's goods and services as both business and consumer products, most likely your company's sales will increase as you build relationships...
-
One pound mass of air $(\mathrm{MW}=29)$ at sea level and $70^{\circ} \mathrm{F}$ is contained in a balloon, which is then carried to an elevation of $10,000 \mathrm{ft}$ in the atmosphere. If the...
-
You must size an orifice meter to measure the flow rate of gasoline ( $\mathrm{SG}=0.72)$ in a $10 \mathrm{in}$. ID pipeline at $60^{\circ} \mathrm{F}$. The maximum flow rate expected is $1000...
-
Calculate the atmospheric pressure at an elevation of $3000 \mathrm{~m}$, assuming (a) air is incompressible, at a temperature of $59^{\circ} \mathrm{F}$; (b) air is isothermal at $59^{\circ}...
-
If $100 million is deposited into the banking system, what would happen to the money supply, according to the simple deposit multiplier, if the required reserve ratio is 4%? What if the required...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
Show how each of the following compounds could be converted to benzoic acid: (a) (b) (c) (d) (e) Benzyl alcohol (f) Benzaldehyde Br
-
Two unsymmetrical anhydrides react with ethylamine as follows: Explain the factors that might account for the formation of the products in each reaction. O C NH2 O C CF CF O- NH
-
Starting with 1-naphthol, suggest an alternative synthesis of the insecticide Sevin to the one given in Section 17.9A.
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
To learn more about the field of HR, go to the HR Career Advice site (http://www.hrcareeradvice.com/Links to an external site.) and read the page "Human Resources Overview." answer the following...
-
Cobe Company has already manufactured 23,000 units of Product A at a cost of $25 per unit. The 23,000 units can be sold at this stage for $420,000. Alternatively, the units can be further processed...

Study smarter with the SolutionInn App


