For each of the following pairs of compounds, determine the relationship between the two compounds. a. b.
Question:
a.
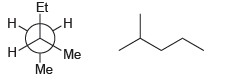
b.
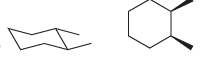
Transcribed Image Text:
Et Н н Me Н Me
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a 3methylpentane and 2methy...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following pairs of compounds undergoes a Bronsted acid-base reaction for which the equilibrium lies to the right. Give the products of each reaction, and identify the acid, the base, the...
-
In each of the following pairs of compounds one is chiral and the other is achiral. Identify each compound as chiral or achiral, as appropriate. a. b. c. d. CICH CHCH OH and HOCH CHCH,OH CI OH CH,CH...
-
In each of the following pairs of compounds choose which one will react faster with the indicated reagent, and write a chemical equation for the faster reaction: (a) Toluene or chlorobenzene with a...
-
A new out-of-state client, Robert Ball, has asked you to prepare a Form 709 for a large gift he made in 2013. When you request copies of any prior gift tax returns he may have filed, he responds,...
-
Two examples of the marketization of society today that Sandel discusses are cash for grades and pay for getting around long lines. What, if anything, is wrong with these practices? Beyond what...
-
The city council approved the following budget for 20X1: Expenditures: City Manager Police Department Fire Department. Streets and Roads Revenues: Property Taxes Fines and Fees Miscellaneous Prior...
-
The accounts of Highland Consulting, Inc., follow with their normal balances at August 31, 2010. The accounts are listed in no particular order. Requirements 1. Prepare the companys trial balance at...
-
Stella Company issues $3 million, 10-year, 9% bonds at 96, with interest payable on July 1 and January 1. The straight-line method is used to amortize bond discount. (a) Prepare the journal entry to...
-
Let z =-2+4i and z2 = 3 + 2i. Perform the indicated operations and write the solutions in the form a + bi. Show your work. 8. Z- Z2 9. 22 10. Zl 11. Z (conjugate) 12. Let z =-1-3i, Z2 = 2 + 3i, Z3 =...
-
Please solve this problem using C language Hacker Industries has a number of employees. The company assigns each employee a numeric evaluation score and stores these scores in a list. A manager is...
-
Draw a Fischer projection for each of the following compounds, placing the CO2H group at the top. a. b. c. . .
-
It is possible for a compound to be chiral even though it lacks a carbon atom with four different groups. For example, consider the structure of the following compound which belongs to a class of...
-
What role does database marketing play in direct-response advertising?
-
A centrifuge with a radius of $2 \mathrm{ft}$ and a length of $1 \mathrm{ft}$ has an overflow weir located $1 \mathrm{ft}$ from the centerline. If particles with $S G=2.5$ and diameters of $10 \mu...
-
The "yield stress" for carbon steel is 35,000 psi, and the "working stress" is one-half of this value. What schedule number would you recommend for a pipe carrying ethylene at a pressure of $2500...
-
A dilute aqueous slurry containing solids with a diameter of $20 \mu \mathrm{m}$ and $S G=1.5$ is fed to a centrifuge rotating at $3000 \mathrm{rpm}$. The radius of the centrifuge is $18...
-
Determine the weight of $1 \mathrm{~g}$ mass at sea level in units of: (a) dynes (b) $\mathrm{lb}_{\mathrm{f}}$ (c) $\mathrm{g}_{\mathrm{f}}$ (d) poundals
-
A gas well contains hydrocarbon gases with an average molecular weight of 24 , which can be assumed to be an ideal gas with a specific heat ratio of 1.3. The pressure and temperature at the top of...
-
If $100 million is withdrawn from the banking system, what would happen to the money supply, according to the simple deposit multiplier, if the required reserve ratio is 4%? What if the required...
-
Fill in each blank so that the resulting statement is true. 83 + 103 = ______ .
-
Given here are the 1H NMR spectra and carbonyl IR absorption peaks of five acyl compounds. Propose a structure for each. (a) (b) (c) (d) (e) OgH 1404 H NMR Spectrum Triplet 1.2(6H) Singlet 2.5(4H)...
-
Compound X (C7H12O4) is insoluble in aqueous sodium bicarbonate. The IR spectrum of X has a strong absorption peak near 1740 cm-1, and its broadband proton-decoupled 13C spectrum is given in Fig....
-
Compound Y (C8H4O3) dissolves slowly when warmed with aqueous sodium bicarbonate. The IR spectrum of Y has strong peaks at 1779 and at 1854 cm-1. The broadband proton-decoupled 13C spectrum of Y...
-
Calculate the number of units to be produced in May if your desired ending inventory is 40% of the next month's sales, beginning finished goods inventory for May was 36,000 units, May's sales are...
-
During the first 10 years of DHS , what were some successes/strengths and failures/weaknesses of the new agency. How efficient and effective is it to have so many functions housed with one agency?...
-
There does not seem to be much to the Organizational Test for tax-exempt status. Why is it so important that the formation documents of the organization have particular language in them?

Study smarter with the SolutionInn App


