Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3
Question:
Explain which of the reaction would provide a better synthesis of3-hexanone.
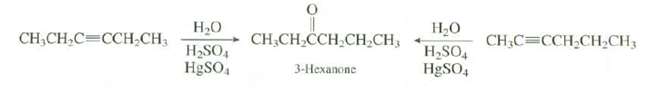
Transcribed Image Text:
Н.О Н.О CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The left synthesis starting with 3hex...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which route would provide a better synthesis of theseethers: CH3 CH, CH3 CHI + CH,CO a) CH.O + CH,CCI CH3 CH,COCH3 H3 CH3 CH, CH,OCHCH,CH; CH,Br CH,0 Br + CH;CHCH,CH3 b) + CH;CHCH,CH,
-
Explain which of these reactions would provide a better synthesis of2-pentene: Br CH,OH CH,CH,CHCH,CH, + CH;0 CH,CH=CHCH,CH3 Br CH, CH CH=CHCH CH, CH,CHCH,CH,CH; + CH,0
-
Explain which of the following approaches to strategy formulation is more likely to generate economic profits: (a) Evaluating external opportunities and threats and then developing resources and...
-
What is the common name for the following structure? CH3 CH3 -C-Br 1 CH3 Isobutyl bromide Osec-Butyl bromide O Neobutyl bromide O Isopropyl methyl bromide t-Butyl bromide
-
Ferguson Gas wants to move its sales order system online. Under the proposed system, gas stations and other merchants will use a secure site to check the availability and current price of various...
-
ANES2008* Estimate with 95% confidence the number of American adults who do not have health insurance (HEALTH: 1 = Yes, 5 = No).
-
Duck Petroleum hires a drilling contractor to drill a well to the depth of 8,000 feet at a cost of $300,000. The $300,000 cost includes $5,000 for surface casing. Any drilling to be completed after...
-
The 23,000-seat City Coliseum houses the local professional ice hockey, basketball, indoor soccer, and arena football teams, as well as various trade shows, wrestling and boxing matches, tractor...
-
Required information P4-7 Recording Adjusting and Closing Entries and Preparing a Balance Sheet and Income Statement Including Earnings per Share LO4-1, 4-2, 4-4 Skip to question [The following...
-
Comparative financial statement data for Cool Brewery Ltd. and Northern Beer Ltd., two competitors, follow (amounts in thousands): Required a. Prepare a common-size analysis for 2020 for Cool Brewery...
-
Show the products of thesereactions: 1) Hg(O,CCH,) . 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH b) a) . H SO4 . H2SO4 d) HgSO, H9SO,
-
Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
-
An unmanned monitoring system uses high-tech video equipment and microprocessors to detect intruders. A prototype system has been developed and is in use outdoors at a weapons munitions plant. The...
-
For small perturbation supersonic flow around a thin airfoil, write the linearized decomposition of the surface pressure coefficient?
-
In transonic airfoil flow, how to control the strength and stability of shock wave on the upper airfoil?
-
What is the concept of entropy and why is dq/T a function of state?
-
Explain the physical phenomenon that the total temperature of a system remains unchanged and the total pressure decreases. Explain a physical phenomenon in which the total temperature and total...
-
Please point out the main measures to increase the drag divergence Mach number?
-
In the absence of a more coherent test, truth and fairness can only really be measured by using the formal requirements contained in the Companies Act 1985 and the FRSs and SSAPs as a series of...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Consider a solution that contains both C 5 H 5 N and C 5 H 5 NHNO 3 . Calculate the ratio [C 5 H 5 N]/[C 5 H 5 NH + ] if the solution has the following pH values: a. pH = 4.50 b. pH = 5.00 c. pH =...
-
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity, and explain yourordering. NH2 Histamine H.
-
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain. Oxazole N:
-
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount. :0: H2SO4 NH2 NH2 R.
-
The charge on your rent-to-own washer is $40 per month. After 32 months, you get to keep the washer. How much does the washing machine end up costing you? Cost of washing machine
-
Prepare horizontal analysis on income statement below: Bristol Myers Squibb Co. (NYS: BMY) Report Date 2020 2019 2018 Net product sales 41,321 25,174 21,581 Alliance & other revenues 1,197 971 980...
-
P8-5A. Using the ledger balances and additional data provided, do the following for Meyer Lumber of Hinton for the year ended December 31, 2022: a. Prepare the worksheet. b. Prepare the income...

Study smarter with the SolutionInn App


