Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,,
Question:
Show the products of thesereactions:
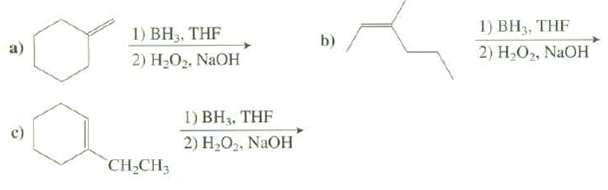
Transcribed Image Text:
1) BH3, THF 1) BH3. THF 2) Н.О. NaOH b) 2) Н,О, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a OH o b ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
As CEO of Aqua Marine, Carrie Easton knows it is important to control costs and to respond quickly to changes in the highly competitive boat-building industry. When Rose Consulting proposes that Aqua...
-
Hazardous materials are constantly being around the country. To help determine how dangerous these events are a statistics practitioner recorded the distances of a random sample of trucks, trains,...
-
The Kincaid Oil Company has unproved property costs of $40,000 at January 1, 2016. During 2016, Kincaid incurred $400,000 drilling costs on Lease A. An 8%, $500,000 note is outstanding during the...
-
CCN and Act Media provided a television channel targeted to individuals waiting in super-market checkout lines. The channel showed news, short features, and advertisements. The length of the program...
-
Depreciation Method annual depreciation expense for year 1 annual depreciation expense for year 2 units-of-activity straight-line double-declining- balance
-
1. Why are there more workplace injuries among those aged 16 to 25? 2. By law, workplace safety is the responsibility of the employer and employee. Why have nearly all provinces created courses in...
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Determine whether the statement is true or false. If it is true, explain why. If it is false, explain why or give an example that disproves the statement. The work done by a conservative force field...
-
What is the Mach wave when passing through the compression angle? Why does the Mach wave line converge?
-
Write the continuous equation, tangential momentum equation, and normal momentum equation that pass through the Mach wave line?
-
A two dimensional airfoil is placed in the air so that its lowest pressure point appears on the lower surface. The pressure coefficient at this point is 0.782 when far field Mach number is 0.3. Try...
-
At low speed Incompressible flow, the pressure coefficient at a given point on the wing is 0.54. When the free flow Mach number is 0.58, by use of (1) Prandtl-Glauert law (2) Karman-Qian law (3)...
-
Under the linearized condition, please write down the formulation of the definite solution problem of the velocity potential function of supersonic flow? The linearized expression of the wall...
-
Before the publication of SAS 600, the standard audit report read as follows: Compare and contrast this report with the current version. Do you consider the differences to be significant? Which...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
Calculate the mass of sodium acetate that must be added to 500.0 mL of 0.200 M acetic acid to form a pH = 5.00 buffer solution.
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH 3,5-Dimethylisoxazole
-
Account for the fact that p-nitro aniline (pKa = 1.0) is less basic than m-nitro aniline (pKa = 2.5) by a factor of 30. Draw resonance structures to support your argument. (The PKa values refer to...
-
Project Risk Management Plan General Project Name: Project Sponsor: Project Manager: Risk Environment [Describe how your project supports your company's strategic plan and why the project is...
-
Current Attempt in Progress Blue Spruce Corp. issued $571,000 of 5-year, 5% bonds at 96 on January 1, 2022. The bonds pay interest annually. (a1) Your answer is correct. Prepare the journal entry to...
-
Net income Year 1 $24,000 Year 2 $34,000 Year $67,000 $57,000 $134,000 Compute the machine's payback period (ignore taxes). (Round payback period answ Computation of Annual Depreciation Expense Year...

Study smarter with the SolutionInn App


