How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
Question:
How would you carry out the following transformations?
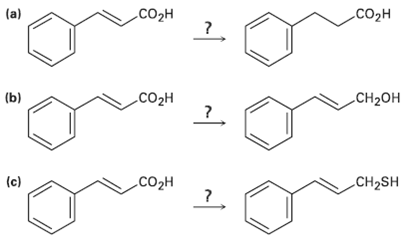
Transcribed Image Text:
Coорн .CO2H (a) (b) Coон CH2он Cоон (c) CH2SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
In these compounds you want to reduce some but not all of the functional group...View the full answer

Answered By

PU Student
cost accounting
financial accounting
auditing
internal control
business analyst
tax
i have 3 years experience in field of management & auditing in different multinational firms. i also have 16 months experience as an accountant in different international firms. secondary school certification.
higher secondary school certification.
bachelors in mathematics.
cost & management accountant
4.80+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? "Co CH2 C Lc
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
On January 1, 2014, Iron Mountain Ski Corporation purchased a new snow-grooming machine for $50,000. The machine is estimated to have a 10-year life with a $2,000 salvage value. What journal entry...
-
Describe the market requirements and product characteristics in global competition.
-
Green Garden Supplies makes ground covers to prevent weed growth. During May, the company sold 44,300 rolls and recorded the following cost data: Requirements 1. Compute the price and efficiency...
-
Your company is evaluating a new project, and you've estimated that the cost of capital for the project will be 8 percent. Assume that you have also projected the following cash flows for the...
-
While analyzing data, an investigator treats each score as if it were contributed by a different subject even though, in fact, scores were repeated measures. What effect, if any, would this mistake...
-
A Company has a note payable for $300,000 on January 31, 2014. Starting on February 1, 2014, the company is required to pay $75,000 on the note each month. The first payment is on February 1, 2014,...
-
A young couple, Eve and Steven, want to divide their main household chores (marketing, cooking, dishwashing, and laundering) between them so that each has two tasks but the total time they spend on...
-
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (b) CH |(a) (c) H CHCH2CH2H2H2 C CHCH-CH
-
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities. (a) 2-Methyl-2-propanol (b) 1-Ethylcyclohexanol (c) 3-Phenyl-3-pentanol...
-
Why do you think cost drops in importance as an objective right after the formation stage?
-
Simple Things completed the following selected transactions during 2015 & 2016 2015 Dec. 31 Estimated that uncollectible-account (bad-debt) expense for the year was 3% of credit sales of $1,000,000...
-
What is often a reason for initial pay disparity between men and women? Also What are the reasons for the disparity between the wages of men and women?
-
On January 1, 2015, Matadors, Inc. purchased $100,000 of five-year, 8% bonds when the effective rate of interest was 10%, paying $92,277.Interest is to be paid on July 1 and December 31. Prepare the...
-
Given the trial balance: 1) prepare a multi-step income statement 2) a statement of retained earnings 3) prepare a classified balance sheet Cash Accounts Receivable, net Inventory, December 31...
-
You are a director/supervisor of a Regulated Education and Care Service. You have been asked to identify the learning and development needs of your organisation. After speaking to management, you...
-
a. What precautions are taken by the embezzler to prevent detection of lapping? b. What tests can the auditor use to detect lapping?
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
We have learned that the enthalpy of vaporization of a liquid is generally a function of temperature. If we wish to take this temperature variation into account, we cannot use the ClausiusClapeyron...
-
Write structural formulas for all of the stereoisomers of 1, 3-dimethylcyclopentane. Label pairs of enantiomers and meso compounds if they exist.
-
Write formulas for all of the isomers of each of the following. Designate pairs of enantiomers and achiral compounds where they exist. (a) 1-Bromo-2-chlorocyclohexane (b) 1-Bromo-3-chlorocyclohexane...
-
Write bond-line three-dimensional formulas for the starting compound, the product, and all of the intermediates in a synthesis similar to the one just given that relates the configuration of...
-
Data governance provides Select one: A. Definitions, standards and procedures for how data is used. B. The internal data entry processes needed to capture accounting transactions. C. A road map that...
-
When it comes to cost allocation, there are various best practices that can be used. One is to thoroughly record the allocation process. This helps to guarantee that costs are allocated consistently...
-
Contractor Proposal Cost Calculation Table COST ELEMENT CHANGE ($) Material $5,000.00 Subcontracts $3,000.00 Direct Labor $2,000.00 Indirect Expenses (Fringe Only) (28.33%) $566.60 Other Direct...

Study smarter with the SolutionInn App


