Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
Question:
Predict the products of the following reactions:
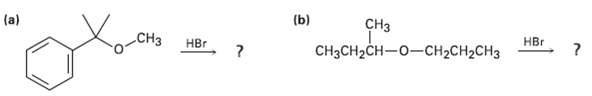
Transcribed Image Text:
(a) (b) CHз CH3CH2CH-0-CH2CH2CH3 CHз НЕr HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a First notice the substitution pattern of the ether Bo...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
Which is found in the java.sql package that come with the standard JDK? A. Only DerbyDriver B. Only MySqlDriver C. Only OracleDriver D. DerbyDriver, MySqlDriver, OracleDriver E. Only DerbyDriver and...
-
What are some of the issues with using financial metrics to evaluate IT?
-
Estimating the cost of old infrastructure is one of the most challenging problems facing governments as they implement Statement No. 34. Fortunately the GASB permits an alternative to reconstructing...
-
If someone had paid \($3\) for a put with E = \($37,\) and the stock was selling for \($40\) at termination of the put, what would be this person's profit on the put? a. -$3 b. -$2 c. $0 d. $3
-
International Accounting Standard No. 16 provides companies the option of valuing property, plant, and equipment at either historical cost or fair value. If fair value is selected, then the property,...
-
All answers must be entered as a formula.
-
New West Company recently hired a new accountant whose first task was to prepare the financial statements for the year ended December 31, 2021. The following is what he produced: The owner of the...
-
Rank the following halides in order of their reactivity in the Williamson synthesis: (a) Bromo ethane, 2-bromopropane, bromo benzene (b) Chloro ethane, bromo ethane, 1-iodopropene
-
Write the mechanism of the acid-catalyzed cleavage of tert-butyl cyclohexyl ether to yield Cyclohexanol and 2-methylpropene.
-
Fatty acid molecules can also align to form a bilipid layer that extends in three dimensions. Shown below is a cross section of this structure. This is similar to the micelle shown in Figure 16.23,...
-
Identify the areas that you have power, due to dominant group membership Pick any ONE component of the ADDRESSING model where you have membership in the dominant group and answer the following...
-
Describe one static priority and one dynamic priority real-time scheduling algorithm. You can use these data in answering the question . You should discuss the issue of admission control, and comment...
-
Evidence that participating in the discussion board deepened your understanding of the course material. How do you think your posted messages brought in new material, ideas, or personal experiences...
-
Let S(t) be the time-t price of a nondividend-paying stock. The expected appreciation rate is 10%, and the volatility is 30%. The current stock price is 20. (a) Find the equal-tailed 90% lognormal...
-
1. Highlight three (3) key personal learnings from the information you reviewed. 2. How will you use this information/tips/strategy to help you increase your employability, either in your job search...
-
a. What agency establishes audit standards for government audits? b. What is the relationship between GAGAS and GAAS?
-
Question 2 For an n x n matrix A = form) via (aij)
-
Redo Problem 4.11 using Aspen Plus. Problem 4.11 Steam is produced at 70 bar and some unknown temperature. A small amount of steam is bled off just before entering a turbine and goes through an...
-
Suggest a possible structure for a compound with the formula C7H12O that has the following IR spectrum and explain yourreasoning: 80 40 20 1718 cm 0- 500 1000 2000 1500 2500 3000 3500 4000 Wavenumber...
-
Suggest a possible structure for a compound with the formula C9H10O that has the following IR spectrum and explain yourreasoning: 100 - 50 1706 em- 4000 3500 3000 2500 2000 1000 500 1500 Wavenamber...
-
Forensic laboratories often have to identify various illicit drug samples. Explain how IR spectroscopy could be used to help distinguish between morphine and heroin. CH3CO, . NH NH CH,CO" ...
-
Data for January for Bondi Corporation and its two major business segments, North and South, appear below. Sales revenues, North Variable expenses, North $ 561,000 $ 325,500 Traceable fixed expenses,...
-
Lindon Company is the exclusive distributor for an automotive product that sells for $48.00 per unit and has a CM ratio of 30%. The company's fixed expenses are $324,000 per year. The company plans...
-
St. Germaine Health Center provides a variety of medical services. The company is preparing its cash budget for the upcoming third quarter. The following transactions are expected to occur: (Click...

Study smarter with the SolutionInn App


