Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry
Question:
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant.
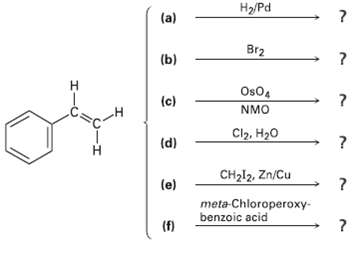
Transcribed Image Text:
H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e) meta-Chloroperoxy- benzoic acid (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a b O c I CCH CCH HIC CCH HPd B...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Predict the products of the following ozonolysis reactions. (a) (b) (c) (1) O2 (2) Me2S (1) 03 (2) Me S (1) O3 (2) Me S
-
Smart housing Inc. is negotiating a deal to build a house. The owner wants to start in early spring when the weather begins to moderate and build through the summer into the fall. The completion time...
-
High school students across the nation compete in a financial capability challenge each year by taking a National Financial Capability Challenge Exam. Students who score in the top 20 percent are...
-
Differentiate between the spot exchange rate and the forward exchange rate.
-
Discuss how you benefit when interest is compounded monthly as opposed to annually.
-
Revenues, production, and purchases budgets. The Suzuki Co. in Japan has a division that manufactures two-wheel motorcycles. Its budgeted sales for Model G in 2010 are 900,000 units. Suzukis target...
-
21 2 points eBook Thornton Company sells lamps and other lighting fixtures. The purchasing department manager prepared the following inventory purchases budget. Thornton's policy is to maintain an...
-
Champion Stores are in trouble. Although the company is a major regional player with hundreds of stores in the upper Midwest, a sharp decline in the regions manufacturing economy has put the company...
-
From what alkene was the following 1, 2-diol made, and what method was used, epoxide hydrolysis or OsO4?
-
Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases. H (b) CH CH3 /Pd CH2H2H2CH H2/Pd (d) CH Br CHH2CHCH (c) Br2 HCI ,...
-
Write a balanced equation to show what happens when CaBr 2 dissolves in water. Use the (aq) symbol when necessary.
-
Gilly is the PFO of Petro, Incorporated. This year, Gilly\'s compensation package was $ 7 3 8 , 0 0 0 , which included $ 6 1 5 , 0 0 0 salary and an accrued, unfunded liability to pay the $ 1 2 3 , 0...
-
What are the key factors that influence enzyme stability and how do these factors play a role in the design of industrial enzymes ?
-
Capacity planning that involves hiring, layoffs, some new tooling, minor equipment purchases, and subcontracting is considered as which one of the following planning horizons?
-
Find four occupations that you think fit the four categories: nonroutine manual jobs; routine manual jobs; routine cognitive jobs and nonroutine cognitive jobs. For each, what is the growth rate in...
-
A proton ( mass 1 , 6 1 0 - 2 7 ???? ???? ) collides with a neutron ( with essentially the same mass as the proton ) to form a particle called a deuteron. What is the velocity of the deuteron, if...
-
Find the equivalent mechanisms with turning pairs for the mechanisms shown in Figs.1.23 (a) to (d).
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
One of these substances is a liquid at room temperature. Which one? a) CH 3 OH b) CF 4 c) SiH 4 d) CO 2
-
Ethyl bromide (0.IM) and HBr (0.1 M) are allowed to react in aqueous THE with 1 M sodium cyanide (Na+ -CN). What products are observed? Arc any products formed more rapidly than others? Explain.
-
What is the expected substitution product (including its stereochemical configuration) in the SN2 reaction of potassium iodide in acetone solvent with the following compound? (D = 2H = deuterium, an...
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
Tyler believes that some laws passed by his state's legislature are bad, and he refuses to comply with them. He believes a universal law above all others that grants rights to all people and that it...
-
Organizational commitment increases the likelihood that an individual will respond to a negative work event with loyalty or neglect. voice or exit. loyalty or voice. O exit or loyalty.
-
In the context of decision-making, satisficing refers to Selecting options at random without any thought O Always choosing the first available option O Avoiding decisions to reduce cognitive load...

Study smarter with the SolutionInn App


