Name each of the following cyclic alkanes. (a) (b) CH3-CH-CH3 CH3
Question:
Name each of the following cyclic alkanes.
(a)
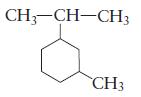
(b)
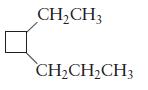
Transcribed Image Text:
CH3-CH-CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a The sixcarbon cyclohexane ring is numbered as follows CH3CHCH3 6 CH...View the full answer

Answered By

Gaurav Bhandari
I have been a teaching assistant for 2 years now and the feedback which i have received from students is very good. They have always praised me for my quality to clear their doubts in the simplest of manners and for my friendly nature so that they never hesitate in asking me all their doubts.
Even though i am in the final year of my graduation at one of India's premier engineering colleges it has always been my dream to be a teacher because i really like to share my knowledge as much as i can and help students achieve their goals in life. I hope i can fulfill my dream through solutionInn.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name each of the following cyclic alkanes, and indicate the formula of the compound. a. b. c. CHCH3 CH3 CH3 CCH3 CH3 CH3 CH3 CH2CH2CH3 CH3
-
Name each of the following cyclic alkanes and indicate the formula of the compound.
-
Each of the following alkanes reacts with chlorine to give a single monochloro substitution product. On the basis of this information, deduce the structure of each alkane. (a) C5H10 (b) C8H18
-
A fruit vendor professes to sell fruits at the cost price, but uses false weights. He gains 40% in this manner. What weight does he substitute for one kilogram?
-
Use the properties of integrals to verify the inequality without evaluating the integrals.
-
Compute the following limits. x + zx lim xx x - 1
-
Purple Company has a current ratio of 2.2 on December 31. On that date its current assets are as follows: Purple Company's current liabilities at the beginning of the year were \(\$ 195,000\) and...
-
(a) Prepare general ledger and subsidiary ledger entries to record the following transactions of the City of Ann Arbor, Michigan, Community Television Network Special Revenue Fund for the year ended...
-
A study of the consultants in a particular industry has determined that the standard deviation of the hourly fee for the consultants is $24. A random sample of 80 consultants in the industry has a...
-
Name each of the following molecules. (a) (b) H CH3CHCH CH3 C=C CH3 H
-
Determine the structure for each of the following compounds. a. 4-ethyl-3,5-dimethylnonane b. 4-tert-butylheptane
-
A banker can always be trusted since they are in a position of customer confidence. T or F
-
Examine the picture of tree leaves below: Public Domain Which description best explains the distortion of color at the bottom of the leaves in the image? Dispersion of light entering the camera lens...
-
River Runs Railroad decided to use the high-low method and operating data from the past six months to estimate the fixed and variable components of transportation costs. The activity base used by...
-
Question 2 a) A consumer has utility function U(, ) = min{+4, +42). 1. Draw the indifference curve for U(,)=20 and shade the area where U(.2) 2 20. 2. For what value of p/P2 will the unique optimum...
-
Below is a graph with two vectors shown on it. Add them, and then draw the resultant vector on the graph in standard position. 10+ 9 8 7 6 5 4 Clear All Draw: 3 2 1 10 -9 -8 -7 -6 -5 -4 -3 -2 -1 -H...
-
A Hall measurement of a semiconductor with a sheet resistance of 200 Ohm/ was performed in 0.5 Tesla, and the Hall voltage is 13 V with a Hall current of 1 A. What are the mobility and sheet carrier...
-
What is defined as the likelihood of disruption that would impact the ability of a company to continuously supply products or services?
-
Define deferred revenue. Why is it a liability?
-
At 500C, K c = 0.061 for N 2 (g) + 3 H 2 (g) 2 NH 3 (g). If analysis shows that the composition of the reaction mixture at 500C is 3.00 mol L 1 N 2 , 2.00 mol L 1 H 2 , and 0.500 mol L 1 NH 3 , is...
-
(a) What is the standard Gibbs free energy of the reaction CO(g) + H 2 O(g) CO 2 (g) + H 2 (g) when K = 1.00? (b) From data available in Appendix 2A, estimate the temperature at which K = 1.00. (c)...
-
The following plot shows a system composed of the gaseous compounds A and B in a rigid, constant-volume flask. The system was initially at equilibrium, then a change occurred. (a) Describe the change...
-
Contesta todo con tus propias palabras . Establecer la ecuacin del modelo, la cual deber de incluir los siguientes datos: Una variable explicada (Yt). Dos variables explicativas (X1 y X2). Dato del...
-
P 2 . 1 2 An investor buys 2 0 0 shares of stock selling at $ 8 0 per share using a margin of 6 0 % . The stock pays annual dividends of $ 1 per share. A margin loan can be obtained at an annual...
-
Scott buys and sells a weekly magazine. The weekly demand for the magazine is approximately normally distributed with a mean of 25 and a standard deviation of 10. Each copy of the magazine costs $4...

Study smarter with the SolutionInn App


