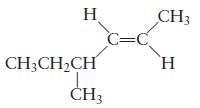
Name each of the following molecules. (a) (b) H CH3CHCH CH3 C=C CH3 H
Question:
Name each of the following molecules.
(a)
(b)
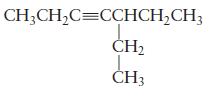
Transcribed Image Text:
H CH3CHCH CH3 C=C CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The longest chain which contains six carbon atoms is number...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2.5 ? solve the equation cx+ly +4=1, subject to the initial condition 4= Sinx, on y=x+x. x>0.
-
Two parallel plates, each charged equally and oppositely to the other, are separated by 2.2500 cm. A proton is let go from rest at the positive plate's surface and, at the same time, an electron is...
-
Name each of the following molecules in accord with the IUPAC system of nomenclature. a. b. c. d. e. f. g. h. i. Cl OH Br Cl HO CF3 Cl Cl CH3O OCH3
-
Let R denote the set of continuous functions with domain R (as in calculus). Then R is a commutative ring with identity. The additive identity OR is the zero function (that is, the function h(x)...
-
Use Property 8 to estimate the value of the integral. 56. V + I dx 55. dx 58. (x - 3x + 3) dx tan x x 57. sin'r dx 59. 60. Je/4
-
Find the equation of the tangent line to the curve y = x 2 at the point (-2, 4). Sketch the graph of y = x 2 and sketch the tangent line at (-2, 4).
-
Purple Company, whose current assets at December 31 are shown in Exercise E13-6B, had net sales for the year of \(\$ 580,000\) and cost of goods sold of \(\$ 339,000\). At the beginning of the year,...
-
Suppose the cross-price elasticity of demand between goods X and Y is 5. How much would the price of good Y have to change in order to increase the consumption of good X by 50 percent?
-
What policy defines what is and what is not an acceptable activity, practice, or use for company equipment and resources? Question 7 options: Everyday use policy Acceptable use policy Bring your own...
-
For each of the following alcohols, give the systematic name, and specify whether the alcohol is primary, secondary, or tertiary. a. CH3CHCHCH3 b. CICHCHCHOH . CH3 CH3CCH, CH,CH, CH,Br
-
Name each of the following cyclic alkanes. (a) (b) CH3-CH-CH3 CH3
-
Interpret the coefficient of determination for the data on actual and recorded inventory values for Aries Car Parts (using the amended data set from Exercise 16). Does this give you confidence in the...
-
A concave spherical mirror has a radius of curvature of magnitude 21.0 cm. (a) Find the location of the image for the following object distances. (If there is no image formed enter "NONE".) object...
-
Determine the current flowing through R2 for the circuit if V=413 Volts, R1=2,856 Ohms, and R2=3,7930hms. R13 Your Answer: Answer R2 units
-
This week we will be discussing the patient-physician relationship. Not everyone has a primary care physician they can call "my PCP" although they have the means to do so through insurance. They wait...
-
What is the relationship between social control and the construction of social identity, particularly in terms of how conformity to societal norms and expectations influences individual identity...
-
Use the given die class for the problems below: public class Die { //attributes private int faceValue; // declare an attribute //operations //default constructor public Die() { //faceValue = 2;...
-
Match the following common risks with the appropriate mitigation strategy. Country risks A: Detailed tracking, alternate suppliers Regulatory risk B: Carefully select and monitor suppliers Logistics...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
(a) Calculate K at 25 C for the reaction Br 2 (g) 2 Br(g) from the thermodynamic data provided in Appendix 2A. (b) What is the vapor pressure of liquid bromine? (c) What is the partial pressure of...
-
Let be the fraction of PCl 5 molecules that has decomposed to PCl 3 and Cl 2 in the reaction PCl 5 (g) PCl 3 (g) + Cl 2 (g) in a constant-volume container; then the amount of PCl 5 at equilibrium...
-
At 2500. K, the equilibrium constant is K c = 20. for the reaction Cl 2 (g) + F 2 (g) 2 ClF(g). An analysis of a reaction vessel at 2500. K revealed the presence of 0.18 mol L 1 Cl 2 , 0.31 mol L 1...
-
Broke Benjamin Co. has a bond outstanding that makes semiannual payments with a coupon rate of 5.8 percent. The bond sells for $997.27 and matures in 16 years. The par value is $1,000. What is the...
-
Explain how investors react to changes in a company's dividend payout policy. Discuss what a firm's payout policy indicates about the financial status of a company. Discuss how taxes impact an...
-
Hi, please help with this question. I was able to answer part a but have gotten stuck on B and C. Thank you (Efficiency analysis) The Brenmar Sales Company had a gross profit margin(gross...
The Psychology Of Attachment The Psychology Of Everything 1st Edition - ISBN: 0367896567 - Free Book

Study smarter with the SolutionInn App


