The balanced equation for the reaction of gaseous nitrogen dioxide and fluorine is The experimentally determined rate
Question:
The balanced equation for the reaction of gaseous nitrogen dioxide and
fluorine is
![]()
The experimentally determined rate law is
![]()
A suggested mechanism for this reaction is
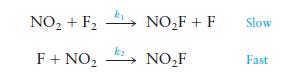
Is this an acceptable mechanism? That is, does it satisfy the two requirements?
Transcribed Image Text:
2NO(g) + F(g) 2NOF(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The first requirement for an acceptable mechanism is that the sum of ...View the full answer

Answered By

Patrick Busaka
I am a result oriented and motivated person with passion for challenges because they provide me an opportunity to grow professionally.
5.00+
38+ Reviews
58+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
3. Calculate and Explain, how much the action described has added to GDP: A company sells 50 computers at a retail price of $1000 apiece and 100 software packages at a retail price of $50 apiece to...
-
The reaction of nitric oxide (NO(g)) with molecular hydrogen (H2(g)) results in the production of molecular nitrogen and water as follows: 2NO(g) + 2H 2 (g) N 2 O(g) + 2H 2 O(g) The experimentally...
-
The dissolved oxygen present in any highly pressurized, hightemperature steam boiler can be extremely corrosive to its metal parts. Hydrazine, which is completely miscible with water, can be added to...
-
Here is the end-of-year account balance information from the accounting records of Jaunty Coffee Company: Sales revenue Cash Cost of goods sold Accounts payable Capital stock Dividends Retained...
-
(a) Use a computer algebra system to draw a direction field for the differential equation. Get a printout and use it to sketch some solution curves without solving the differential equation. (b)...
-
The butterfly curve is given by the following parametric equations: Generate values of x and y for values of t from 0 to 100 with t = 1/16. Construct plots of (a) x and y versus t and (b) y versus x....
-
Lake Company has the following shares outstanding: 20,000 shares of \(\$ 50\) par value, five percent cumulative preferred stock and 80,000 shares of \(\$ 10\) par value common stock. The company...
-
What are the expected value and variance of the following probability distribution? RANDOM VARIABLE X PROBABILITY 1 ................0.05 2 ................0.05 3 ................0.10 4...
-
A $100,000 bond has a term to maturity of one year. There are two coupon interest payments yet to be paid, one due in 6 months and the second to be paid along with the face value when the bond...
-
The gas-phase reaction of chlorine with chloroform is described by the equation The rate law determined from experiment has a noninteger order: A proposed mechanism for this reaction follows: Is this...
-
Butadiene reacts to form its dimer according to the equation 2C 4 H 6 (g) C 8 H 12 (g) The following data were collected for this reaction at a given temperature: a. Is this reaction first order or...
-
List the primary types of physical media in use in telecommunications networks today. Which of these media has the fastest transmission speed? The slowest transmission speed?
-
Solve. 2w219w+31+2=7- 2w
-
Suppose that you are the treasurer of IBM with an extra U.S. $1,000,000 to invest for six months. You are considering the purchase of U.S. T-bills that yield 1.810 percent (that's a six month rate,...
-
What is the price of a European call option on the stock of Company Z when the current stock price is $50, the strike price is $48, the risk free rate is 12% continuously compounded, the standard...
-
Swifty Inc. had beginning inventory of $23,000 at cost and $32,000 at retail. Net purchases were $156,302 at cost and $206,000 at retail. Net markups were $11,300, net markdowns were $7,000, and...
-
5. A woman is saving for retirement in 15 years. She currently has $300,000 in a savings account earning 6% per year. In addition, she is saving $22,000 per year in an account earning 4% per year....
-
A manufacturing facility has five jobs to be scheduled into production. The following table gives the processing times plus the necessary wait times and other necessary de-lays for each of the jobs....
-
Digital Fruit is financed solely by common stock and has outstanding 25 million shares with a market price of $10 a share. It now announces that it intends to issue $160 million of debt and to use...
-
Draw Lewis structures for each of the following species and predict the hybridization at each carbon atom: (a) H 2 CCH ; (b) H 2 CCH 3 + ; (c) H 3 CCH 2 .
-
There are three isomers of difluoroethene, C 2 H 2 F 2 , which differ in the locations of the fluorine atoms. (a) Which of the forms are polar? (b) Which has the largest dipole moment? F F C= 1 C O H...
-
Explain why the lattice energy of lithium chloride (861 kJ mol -1 ) is greater than that of rubidium chloride (695 kJ mol -1 ), given that they have similar arrangements of ions in the crystal...
-
You have the following information about two banks Total Deposits ( $ ) Total Assets ( $ ) sA Bank 1 $ 2 5 0 , 0 0 0 , 0 0 0 , 0 0 0 $ 5 0 0 , 0 0 0 , 0 0 0 , 0 0 0 7 % Bank 2 $ 4 0 , 0 0 0 , 0 0 0 ,...
-
The Hastings Sugar Corporation has the following pattern of net income each year, and associated capital expenditure projects. The firm can earn a higher return on the projects than the stockholders...
-
42 (1 point) The (GAAP) must be followed by publicly owned companies and are changed and refined in response to changes in the environment in which businesses operate. general accounting & auditing...

Study smarter with the SolutionInn App


