The gas-phase reaction of chlorine with chloroform is described by the equation The rate law determined from
Question:
The gas-phase reaction of chlorine with chloroform is described by the equation
![]()
The rate law determined from experiment has a noninteger order:
![]()
A proposed mechanism for this reaction follows:
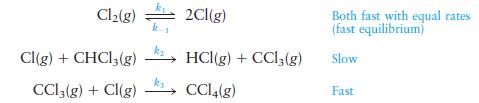
Is this an acceptable mechanism for the reaction?
Transcribed Image Text:
Cl(g) + CHCl3(g) HCl(g) + CCl4(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Two questions must be answered First does the mechanism giv...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name this amine, including (R.S) stereochemistry. bal & stick labels (It is not necessary to use italics in writing compound names.)
-
tive 2.1 Managerial Cost Concepts You reached 100% progress. Keep going in Study Mode or exit the assignment. Major Industries manufactures covers for iPads. One of its most popular products,...
-
Calculate the net sales for 2021 and 2022 Fiscal Years Ended Revenue Cost of sales Gross profit Selling, general and administrative expenses Restructuring charges Operating income Other income...
-
Which of the following is not an example of supervised machine learning? O Clustering O Deep learning O Decision Tree Linear Regression
-
Find the orthogonal trajectories of the family of curves. Use a graphing device to draw several members of each family on a common screen. 27. y = kx? 29. y = (x + k)- 28. x? - y? - k 30. y - ke
-
Develop an M-file function that is passed a numeric grade from 0 to 100 and returns a letter grade according to the scheme: The first line of the function should be function grade =...
-
Minaret, Inc., issued 10,000 shares of \(\$ 50\) par value preferred stock at \(\$ 68\) per share and 12,000 shares of no-par value common stock at \(\$ 15\) per share. The common stock has no stated...
-
Consider a project to supply 60,800,000 postage stamps to the U.S. Postal Service for the next 5 years. You have an idle parcel of land available that cost $760,000 five years ago; if the land were...
-
c . Calculate the firms cash conversion cycle given that annual sales are $ 6 6 0 , 0 0 0 and cost of goods sold represents 9 0 % of sales. Assume a 3 6 5 - day year.
-
The reaction was studied at several temperatures and the following values of k were obtained: Calculate the value of E a for this reaction. 2NOs(g) 4NO2(g) + O(g)
-
The balanced equation for the reaction of gaseous nitrogen dioxide and fluorine is The experimentally determined rate law is A suggested mechanism for this reaction is Is this an acceptable...
-
The financial statements for Nike, Inc., are provided in Appendix E at the end of the text. Determine the free cash flow for the year ended May 31, 2007. Assume that 90% of additions to property,...
-
Define the regression analysis and explain with example FIVE (5) situation regression analysis been conducted. 2 type of regression analysis : - linear analysis multiple analysis
-
Bill's Clothing Company has a cost of debt of 7 percent, a cost of equity of 11 percent, and a cost of preferred stock of 8 percent. The firm has 104,000 shares of common stock outstanding at a...
-
Diageo was conglomerate involved in food and beverage industry in 1997. Initially founded by a merger between Grand Metropolitan and Guinness, the company went to become a significant player in the...
-
15 years ago, Company X issued some 20 year 5% annual coupon bonds that were priced with a yield to maturity of 8% p.a. and had a face value of $1000. What did these bonds sell for when they were...
-
The following information is from Harrelson Inc.'s financial statements. Sales (all credit) were $28.5 million for last year. Sales to total assets Total debt to total assets Current ratio Inventory...
-
The following table contains information regarding jobs that are to be scheduled through one machine: a. What is the first- come, first- served (FCFS) schedule? b. What is the shortest operating time...
-
Determine the volume of the parallelepiped of Fig. 3.25 when (a) P = 4i 3j + 2k, Q = 2i 5j + k, and S = 7i + j k, (b) P = 5i j + 6k, Q = 2i + 3j + k, and S = 3i 2j + 4k. P
-
(a) Consider the hypothetical species HeH. What charge (magnitude and sign), if any, should be present on this combination of atoms to produce the most stable molecule or ion possible? (b) What is...
-
(a) Draw the bonding and antibonding orbitals that correspond to the -bond in H 2 . (b) Repeat this procedure for HF. (c) How do these orbitals differ?
-
Explain why the lattice energy of silver bromide (903 kJ mol -1 ) is greater than that of silver iodide (887 kJ mol -1 , given that they have similar arrangements of ions in the crystal lattice.
-
$882,038 $991,492 $854,426 $750,550 $1,103,426
-
a. Find the present values of the following cash flow streams at a 3% discount rate. Do not round intermediate calculations. Round your answers to the nearest cent. O 1 2 3 4 5 Stream A $0 $100 $400...
-
You are given the following information for Huntington Power Company. Assume the company s tax rate is 2 5 percent. Debt: 3 4 , 0 0 0 5 . 3 percent coupon bonds outstanding, $ 2 , 0 0 0 par value, 2...

Study smarter with the SolutionInn App


