In a reactive environment, chemical A decomposes as follows For a feed stream C A0 = 4
Question:
In a reactive environment, chemical A decomposes as follows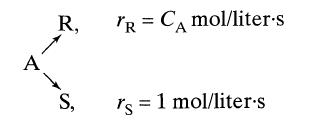
For a feed stream CA0 = 4 mol/ter what size ratio of two mixed flow reactors will maximize the production rate of R? Also give the composition of A and R leaving these two reactors.
Transcribed Image Text:
A R, S, TR = CA mol/liter-s rs 1 mol/liter.s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Substance A in the liquid phase produces R and S by the following reactions: The feed (C Ao = 1.0, C Ro = 0, C so = 0.3) enters two mixed flow reactors in series ( 1 = 2.5 min, 2 , = 10 min)....
-
The first-order reactions are to be run in two mixed flow reactors in series anywhere between 10 and 90C. If the reactors may be kept at different temperatures, what should these temperatures be for...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The summary of financial statements for division A is given below. Statement of Financial Position Statement of Profit or Loss RM'000 RM'000 Non-current assets 1,500 Revenue 4,000 Current assets 600...
-
Data related to the expected sales of 12'' and 16'' pizzas for Pietros Pizza, Inc. for the current year, which is typical of recent years, are as follows: The estimated fixed costs for the current...
-
Surely you welcome the opportunity to join our protective organization. Think of all the money you will lose from broken windows, overturned trucks, and damaged merchandise in the event of your not...
-
Inherent Risk. Consider the following factors: 1 Integrity of management 2 Organization and management structure 3 Nature of the entitys business 4 Industry factors 5 Financial statement accounts...
-
Iridium Corp. has spent $3.5 billion over the past decade developing a satellitebased telecommunication system. It is currently trying to decide whether to spend an additional $350 million on the...
-
A new molding machine is expected to produce operating cash flows of $68,000 a year for 6 years. At the beginning of the project, inventory will decrease by $14,700, accounts receivables will...
-
Consider the parallel decomposition of A of different orders. Determine the maximum concentration of desired product obtainable in (a) Plug flow, (b) Mixed flow. R is the desired product and C Ao =...
-
Reactant A in a liquid either isomerizes or dimerizes as follows: (a) Write (R/A) and ([RI(R + S)]. With a feed stream of concentration C A0 find C R,max which can be formed (b) In a plug flow...
-
The table lists the value y (in dollars) of $1000 deposited in a certificate of deposit at Bank of New York (based on rates currently in effect). Construct a scatterplot and identify the mathematical...
-
The preceding paragraph is an illustration of a sample that was taken from the minutes of a meeting that took place in HEWTO. The purpose of the gathering is to talk about Derek, who expressed...
-
Discuss the two categories of unmanned aircraft. Briefly describe the differences between sensors and payload. Differences between Active and Passive Sensing Differences between Remote and In Situ...
-
Data for the past two years for J&J Corp. are For 2019 units produced were 10,000 and the overhead applied per unit is P15. Actual FOH Variable; 95,000 and Fixed 50,000. Estimated fixed overhead...
-
Use the Black-Scholes option pricing model to price a one-year at the money call option on a stock that is trading at $50 per share, Rf is 5%, annual volatility is 25%. REMEMBER TO USE THE NORMAL...
-
What are the three types of heuristics we depend on to make quick decisions? Which is inherently bad, heuristics, biases, or both? Give one example of a bias coming from affect heuristic. What are...
-
Toshiaki Minamiyama is a successful business executive. Over the years, he has allocated a portion of his large salary to the building of an investment portfolio. He currently has a net worth of...
-
Is times interest earned meaningful for utilities? Why or why not?
-
The first-order, gas-phase, reversible reaction AB+2C is taking place in a membrane reactor. Pure A enters the reactor, and B diffuses out through the membrane. Unfortunately, a small amount of the...
-
With the focus on alternative clean-energy sources, we are moving toward an increased use of fuel cells to operate appliances ranging from computers to automobiles. For example, the hydrogen/oxygen...
-
The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate CH2OHCH2Cl + NaHCO3 (CH2OH)2 + NaCl + CO2 is carried out in a semibatch reactor. A 1.5-molar solution of ethylene...
-
Mind Challenge, Incorporated, publishes innovative science textbooks for public schools. The company's management recently acquired the following two new pieces of equipment. Computer - controlled...
-
FederalWay, Incorporated, is a major department store chain. The dominant portion of the company's business consists of providing merchandise and services to consumers through department stores and...
-
As an auditor for the CPA firm of Hinkson and Calvert, you encounter the following situations in auditing different clients. Splish Brothers Inc. is a closely held corporation whose stock is not...

Study smarter with the SolutionInn App


