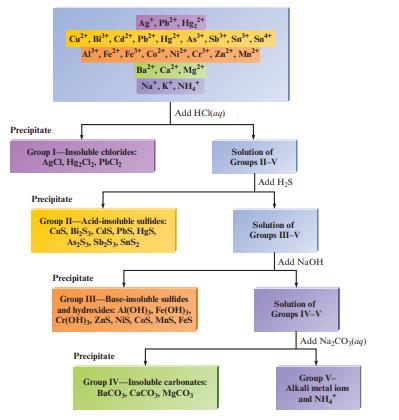
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain
Question:
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain the chemistry involved with each of the four steps in the diagram.
Fig. 15.2

Transcribed Image Text:
Precipitate Ag", Pb²*, Hg: Cu²*, Bi³¹, Cd²*, Pb²+, Hg, As". Sb³*, Sn. AP, Fe, Fe, Co, Ni, Cr³, Zn, Mn Ba, Ca, Mg Na, K, NH, Group 1-Insoluble chlorides: AgCl, HgCl₂, PbCl₂ Precipitate Group II-Acid-insoluble sulfides: CuS, Bi S3, CdS, PbS, H₂S, As2S₁, Sb₂S3, SS₂ Precipitate Add HCl(aq) Precipitate Group III-Base-insoluble sulfides and hydroxides: Al(OH), Fe(OH)3, Cr(OH), ZS, NIS, COS, MuS, Fes Group IV-Insoluble carbonates: BaCO,, CaCO,, MgCO, Solution of Groups II-V Add H₂S Solution of Groups III-V Add NaOH Solution of Groups IV-V Add NaCO3(aq) Group V- Alkali metal ions and NH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
In step one the cations react with HCl to form corresponding salts Salts of AgCl 2 HgC...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another. Explain the chemistry involved in the various steps of the figure. Fig. 15.3 Solution of...
-
Devise a procedure for separating a mixture of the four stereoisomers of isoleucine into its four components: (+)-isoleucine, (-)-isoleucine, (+)-alloisoleucine, and (-2)-alloisoleucine (Problem 31)....
-
Chromatography (Figure 1.14) is a simple but reliable method for separating a mixture into its constituent substances. You have a mixture of two vegetable dyes, one red and one blue, that you are...
-
In Exercises 2748, find the open intervals where the functions are concave upward or concave downward. Find any inflection points. f(x) = 2e x2
-
What If the Facts Were Different? Suppose that Gladys had told Clara that she was giving the rings to Clara but wished to keep them in her possession for a few more days. Would this have affected the...
-
Hotwax completed products costing $275,000 and transferred them to finished goods. Prepare its journal entry to record the transfer of units from production to finished goods inventory.
-
For Lundy Company, the relevant range of production is 40-80% of capacity. At 40% of capacity, a variable cost is \($2,000\) and a fixed cost is \($4,000\). Diagram the behavior of each cost within...
-
The Laurenster Corporation is getting into the construction business. A list of activities and their optimistic, most likely, and pessimistic completion times are given in the following table for the...
-
Evaluate and analyze the performance of Cadbury and Nestle, comparing it with a selected range of competitor (s). (a) describe its operating context within its industrial sector with a brief...
-
Suppose that an investor holds a share of Sophia common stock, currently valued at $50. She is concerned that over the next few months the value of her holding might decline, and she would like to...
-
Under what circumstances can you compare the relative solubilities of two salts directly by comparing the values of their solubility products? When can relative solubilities not be compared based on...
-
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists K sp values for several ionic solids. For any of these ionic compounds, you should be able to calculate the...
-
The following data are given for CCl 4 . Normal melting point, -23 C; normal boiling point, 77 C; density of liquid 1.59 g/mL; fus H = 3.28 kJ mol -1 ; vapor pressure at 25 C, 110 Torr. (a) What...
-
Confirm that the below limit meets the conditions to apply l'Hpital's Rule and then solve the limit. Be sure to address these conditions in your explanation. Enter an exact numeric answer. lim In(x)...
-
ACTIVITY 47: Analyzing Communication A Observe a conversation between two people you've never met. Note all six elements of communication: the identity or role of the sender, the content of the...
-
A thin, metallic, spherical shell of radius 23.7 cm has a total charge of 8.03 C placed on it. A point charge of 2.65 C is placed at the center of the shell. What is the electric field E at a...
-
Discuss inventory management techniques to ensure product availability and minimize overstock. Explain how these techniques contribute to cost control and customer satisfaction.
-
What are the unique challenges of resolving conflicts during crisis situations, and how can conflict resolution professionals prepare for and navigate these high-pressure environments?
-
Let x = sodium and y = sugar for the breakfast cereal data in the Cereal data file on the text CD and in Table 2.3 in Chapter 2. a. Construct a scatterplot. Do any points satisfy the two criteria for...
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
Predict the product of the Dieckmann cyclization that occurs when each of the following compounds is treated with sodium ethoxide. (a) (b) (c) OEt LOET Eto OEt
-
When the following compound is treated with sodium ethoxide, two condensation products are obtained, both of which are produced via Dieckmann cyclizations. Draw both products. OEt Eto
-
For each of the following reactions, predict the major product and propose a mechanism for its formation. (a) (b) (c) :? 1) LDA 2) CH3I 1) NaH 2) -CH,Br
-
Sort the following into asset, liability and stockholder equityAccounts payable. . . . . . . Liability Accounts receivable. . . . . Asset Advertising expense. . . . . ? Building. . . . . . . . . . ....
-
In applying the present value rule for a lease classification, the present value of the residual value a. should be included regardless of guaranteed or not. b. should only be included if it is...
-
When it comes to cost allocation, there are various best practices that can be used. One is to thoroughly record the allocation process. This helps to guarantee that costs are allocated consistently...
Biased Technical Change And Economic Conservation Laws 2006th Edition - ISBN: 0387260552 - Free Book

Study smarter with the SolutionInn App


