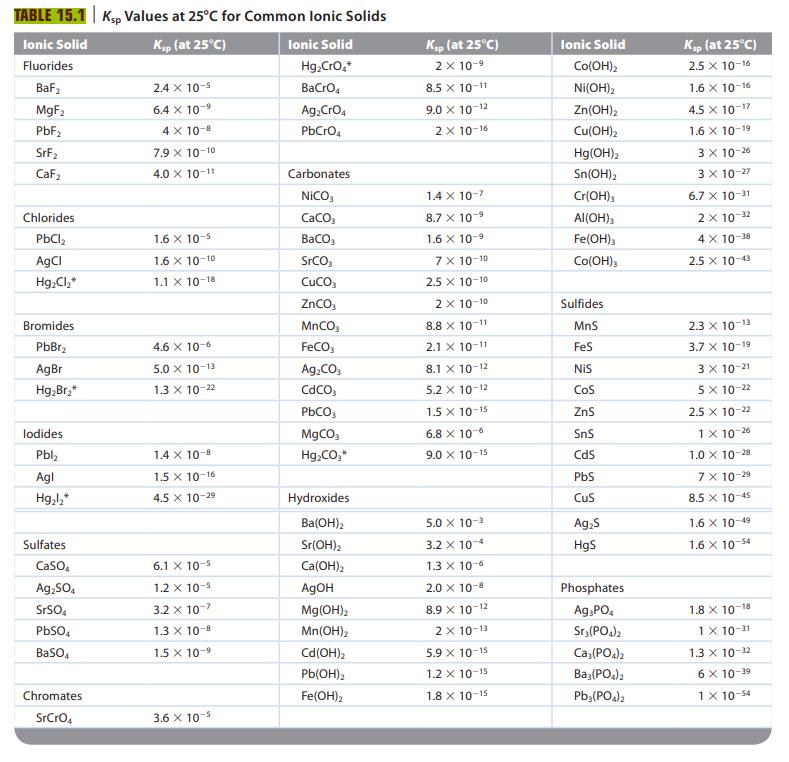
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists K sp values for
Question:
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists Ksp values for several ionic solids. For any of these ionic compounds, you should be able to calculate the solubility. What is the solubility of a salt, and what procedures do you follow to calculate the solubility of a salt? How would you calculate the Ksp value for a salt given the solubility?
Transcribed Image Text:
TABLE 15.1 Ksp Values at 25°C for Common Ionic Solids Ionic Solid Ksp (at 25°C) Fluorides BaF₂ MgF₂ PbF₂ SrF₂ CaF₂ Chlorides PbCl₂ AgCl Hg₂Cl₂* Bromides PbBr₂ AgBr Hg₂Br₂* lodides Pbl₂ Agl Hg₂l₂* Sulfates CaSO4 Ag₂SO4 SrSO4 PbSO4 BaSO4 Chromates SrCrO4 2.4 x 10-5 6.4 x 10-9 4 X 10-8 7.9 X 10-10 4.0 X 10-11 1.6 X 10-5 1.6 X 10-10 1.1 X 10-18 4.6 x 10-6 5.0 X 10-13 1.3 x 10-22 1.4 x 10-8 1.5 X 10-16 4.5 X 10-2⁹ 6.1 x 10-5 1.2 x 10-5 3.2 x 10-7 1.3 X 10-8 1.5 X 10-9 3.6 x 10-5 Ionic Solid Hg₂CrO,* BaCrO4 Ag₂ CrO4 PbCrO4 Carbonates NICO, CaCO3 BaCO3 SrCO CUCO3 ZnCO₂ MnCO₂ FeCO3 Ag₂CO3 CdCO PbCO3 MgCO3 Hg₂CO,* Hydroxides Ba(OH)2 Sr(OH)2 Ca(OH)2 AgOH Mg(OH)2 Mn(OH)₂ Cd(OH)₂ Pb(OH)2 Fe(OH)₂ Ksp (at 25°C) 2 x 10-⁹ 8.5 X 10-11 9.0 X 10-12 2 X 10-16 1.4 x 10-7 8.7 X 10 -9 1.6 × 10-9 7 x 10-10 2.5 X 10-10 2 x 10-10 8.8 x 10-11 2.1 X 10-11 8.1 X 10-12 5.2 x 10-12 1.5 X 10-15 6.8 x 10-6 9.0 X 10-15 5.0 × 10-³ 3.2 x 10-4 1.3 x 10-6 2.0 × 10-8 8.9 X 10-12 2x 10-13 5.9 X 10-15 1.2 X 10-15 1.8 X 10-15 Ionic Solid Co(OH)2 Ni(OH)2 Zn(OH)₂ Cu(OH)₂ Hg(OH)2 Sn(OH)2 Cr(OH)3 Al(OH)3 Fe(OH)3 Co(OH)3 Sulfides MnS FeS NIS COS ZnS SnS CdS PbS CuS Ag₂S HgS Phosphates Ag3PO4 Sr3(PO4)2 Ca3(PO4)2 Ba3(PO4)2 Pb3(PO4)2 Ksp (at 25°C) 2.5 X 10-16 1.6 X 10-16 4.5 X 10-1 1.6 X 10-19 3 x 10-26 3 x 10-27 6.7 X 10-31 2 x 10-32 4 X 10-38 2.5 X 10-43 2.3 X 10-13 3.7 X 10-19 3 x 10-21 5 x 10-22 2.5 x 10-22 1 X 10-26 1.0 X 10-28 7 x 10-29 8.5 x 10-45 1.6 X 10-49 1.6 x 10-54 1.8 X 10-18 1 X 10-31 1.3 x 10-32 6 x 10-39 1 x 10-54
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Lets figure out how soluble BaCrO 4 is Utilizing the supplied K sp value from Table 151 which i...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write a project management plan. we have a template and project description. we need to edit the template(table of contents) with our own ideas. CPSC 8820-01 Project Management Plan Your Unique...
-
Read the article " The Twitter Trap " by Bill Keller carefully. In this article, Keller uses his personal experiences to attract the audience and uses funny examples to help the audience understand...
-
Calculate the solubility product constant for Mg(OH)2 at 25oC. The Gof values (in kJ/mol) are as follows: Mg2+ (aq), 454.8; OH(aq), 157.3; Mg(OH)2(s), 833.7.
-
State whether or not each of the following events would result in a liability being recognised in the accounts at 30 June. 1. Taxes for the year ended 30 June, which are not payable until October. 2....
-
Go to this texts Web site at academic. cengage.com/blaw/clarkson and select Chapter 49. Click on Video Questions and view the video titled Double Indemnity. Then answer the following questions. (a)...
-
Pro-Weave manufactures stadium blankets by passing the products through a weaving department and a sewing department. The following information is available regarding its June inventories: The...
-
Monthly production costs in Obianwu Company for two levels of production are Classify costs as variable. as follows. Indicate which costs are variable, fixed, and mixed, and give the reason for each...
-
Terry and Jessica Benedict invested $6,000 in a savings account paying 4% annual interest when their daughter, Kristi, was born. They also deposited $1,000 on each of her birthdays until she was 18...
-
16- Cash receipts: Cash receipts from customers Sale of cooking range $33,165 400 Total cash receipts $33,565 Cash disbursements: Food and supplies $14,275 City restaurant license, valid September 1,...
-
8.1 Create a one-way data table for profit at different levels of supplier contact in range B22:C33. Ensure that the price in cell C3 is $290 and the advertising budget in cell C5 is $35,000,000 (you...
-
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain the chemistry involved with each of the four steps in the diagram. Fig. 15.2...
-
Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH). b. Rank the four titrations in order of...
-
During the summer between his junior and senior years, James Cook needed to earn sufficient money for the coming academic year. Unable to obtain a job with a reasonable salary, he decided to try the...
-
What are the key challenges and opportunities in cross-cultural negotiation, and how can negotiators leverage cultural differences to build trust and reach mutually beneficial agreements in diverse...
-
Given (a) [f(x) dx = 0 and [f(x). -1 (x) dx f(x) dx = 6, evaluate the following. d (b) (c) L'(x) dx - Lf(x) dx 4f(x) dx LAF (d) 4f(x) dx
-
What if the expected direct labor rate at the beginning of the year was $28 instead of $35? What would the overhead rate be? If required, round your overhead rate answer to one decimal place. New...
-
Light of wavelength 5.40x102 nm passes through a slit of width 0.200 nm. a. Find the width of the central maximum on a screen located 1.50 m from the slit. b. Determine the width of the first-order...
-
Using Human Resources Management as a subject guide for my major, describe in a few words any interesting sources discovered.
-
Using software, analyze the relationship between x = college education and y = percentage single-parent families, for the data in Table 3.6, which are in the U.S. Statewide Crime data file on the...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
Identify the reagents you would use to achieve the following transformation:
-
Propose an efficient synthesis for each of the following compounds using the malonic ester synthesis. (a) (b) (c) (d) (e)
-
Starting with diethyl malonate and using any other reagents of your choice, proposean efficient synthesis for each of the following compounds: (a) (b) (c) .
-
Which organization has brought Canada and US to a common format and valuation in reporting the financial statements for the public companies
-
The process of comparing and checking company records against those shown on a bank statement to determine whether the records are in agreement is called what a) bank reconciliation. b) credit audit....
-
6. Cutting bits in a machinery department 7. Food for a factory cafeteria 8. Workers' compensation insurance in a factory 9. Steel scrap for a blast furnace 10. Paper towels for a factory...

Study smarter with the SolutionInn App


