Which of the following reactions is (are) product-favored at equilibrium? (a) Zn(s) + 1(s) Zn+ (aq)
Question:
Which of the following reactions is (are) product-favored at equilibrium?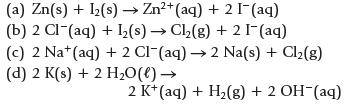
Transcribed Image Text:
(a) Zn(s) + 1₂(s) → Zn²+ (aq) + 2 I¯ (aq) (b) 2 Cl (aq) + ₂(s)→ Cl₂(g) + 2 [¯ (aq) (c) 2 Nat (aq) + 2 Cl (aq) →2 Na(s) + Cl₂(g) (d) 2 K(s) + 2 H₂O(l) → 2 K+ (aq) + H₂(g) + 2 OH-(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The standard Gibbs free energy change can be calculated using the following equation Gf Gfproducts G...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions will have the largest equilibrium constant (K) at 298 K? a) Fe2O3(s) + 3 CO(g) ? 2 Fe(s) + 3 CO2(g) ?G = -28.0 kJ b) It is not possible to determine without more...
-
1) Express the equilibrium constant for the following reaction. P 4 O 10 (s) ? P 4 (s) + 5 O 2 (g) a) K = [O 2 ] -5 b) K = [O 2 ] 5 c) K = d) K = 2) For the reaction of carbon with carbon dioxide to...
-
Write a detailed paper on Trademark Law of the People's Republic of China
-
Use hand calculations to fit the multiple linear regression model 1 y = β0 + β1x1 + β2x2 to the data set in DS 13.6.2. (a) Write down the vector of observed values...
-
Employees expect that all parties will honor their explicit and implicit obligations. Distrust occurs when these obligations are not met or when the parties have different expectations regarding the...
-
The following accounts were taken from the financial statements of Giles Company. ________ Interest revenue ________ Owners capital ________ Utilities payable ________ Accumulated...
-
Under U.S. GAAP, the interest cost associated with borrowings used in the self-construction of a business asset may be capitalized to the balance sheet as part of the self-constructed assets cost...
-
POD Incasements manufactures protective cases for MP3 players. During November, the company's workers clocked 800 more direct labor hours than the flexible budget amount of 25,000 hours to complete...
-
Since East Coast Yachts is producing at full capacity, Larissa has decided to have Dan examine the feasibility of a new manufacturing plant. This expansion would represent a major capital outlay for...
-
You want to set up a series of voltaic cells with specific cell potentials. The Ag + (aq, 1.0 M) | Ag(s) half-cell is one of the compartments. Identify several half-cells that you could use so that...
-
A potential of 0.142 V is recorded (under standard conditions) for a voltaic cell constructed using the following half reactions: (a) What is the standard reduction potential for the anode reaction?...
-
Find the indefinite integrals of the following function. 5/x3
-
Achievable Company produces component A1 for use in one of its electronic gadgets. Normal annual production for the item is 100,000 units. The cost for a 100-unit batch of the part are: Direct...
-
A company's issued share capital consists of $ 2 0 0 , 0 0 0 in 5 0 c ordinary shares issued at a premium of 1 0 c per share. A dividend of 2 0 % is declared. what is the cash payable to shareholders...
-
Recently, Shasta Corporation has decided to play a more-active role in the soda be is aware that the current market price for a can of soda is $2.00. Shasta plans to se soda in its first year and...
-
In the 2nd stage regression used to test the CAPM, researchers reduced the measurement error in betas from the 1ststage regression. What was the source of the measurement error and how did they...
-
how the information management system enhances overall business operations and analysis the role of ICT applications in Operations optimization at PepsiCo
-
Suppose a firm has a tax loss of $5 million in the current period. The firms after-tax discount rate is 10%. Over the preceding 5 years the firm has reported the following taxable income: a. If the...
-
Draw two scatterplots, one for which r = 1 and a second for which r = 21.
-
(a) If gate delays are 5ns, what is the delay of the fastest 4-bit ripple carry adder? (b) If gate delays are 5ns, what is the delay of the fastest 4-bit adder? What kind of adder will it be?
-
Design the correction circuit for a BCD adder that computes Zdigit 0 and C for S 0 . This correction circuit adds 0110 to S 0 if S 0 > 9. This is the same as adding 0AA0 to S 0 , where A = 1 if S 0 >...
-
Consider the following programmable I/O block: Highlight the connections to configure this I/O block as an INPUT pin. Specify the five configuration bits. CONFIGURATION BITS Vcc OUT 3-STATE LATCHED...
-
Assume that Social Security promises you $ 4 3 comma 0 0 0 per year starting when you retire 4 5 years from today ( the first $ 4 3 comma 0 0 0 will be paid 4 5 years from now ) . If your discount...
-
Explain INCO Terms? Can Inco terms help organization to save money? Being a startup in Imports transaction, what are the terms you as Procurement Head for your organization will prefer to avoid...
-
Billy Gatos decided to sell 5,000,000 shares of Microsoft stocks. A broker, fearing that such a transaction would bring down the price of Microsoft shares (and he would be impacted), intercepts the...

Study smarter with the SolutionInn App


