A potential of 0.142 V is recorded (under standard conditions) for a voltaic cell constructed using the
Question:
A potential of 0.142 V is recorded (under standard conditions) for a voltaic cell constructed using the following half reactions: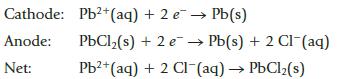
(a) What is the standard reduction potential for the anode reaction?
(b) Calculate the solubility product, Ksp, for PbCl2.
Transcribed Image Text:
Cathode: Anode: Net: Pb2+ (aq) + 2 e PbCl₂(s) + 2 e. Pb²+ (aq) + 2 Cl(aq) → PbCl₂(s) Pb(s) Pb(s) + 2 Cl- (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a To find the standard reduction potential for the anode reaction we can use the following equation ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A cell is constructed using the following half-reactions: (a) What reactions should be observed at the anode and cathode? (b) Calculate the solubility product constant, K sp , for Ag 2 SO 4 . Ag+...
-
Under standard conditions for all concentrations, the following reaction is spontaneous at 25oC. O2(g) + 4H + (aq) + 4Br(aq) 2H2O(l) + 2Br2(l) If [H+] is decreased so that the pH = 3.60, what value...
-
Under standard conditions for all concentrations, the following reaction is spontaneous at 25oC. O3(g) + 2H + (aq) + 2Co2+(aq) O2(g) + H2O(l) + 2Co3+(l) If [H+] is decreased so that the pH = 9.10,...
-
Svengool Inc. financial statements included the following amounts for the current year: Retired bonds $67,000 30,000 Proceeds from collection of note receivable Dividends received 11,000 Acquired...
-
Borders and Amazon.com are competitors in vending books and other consumer items. The two are differentiated to an extent by their marketing strategies. Although Amazon.com relies exclusively on...
-
The trial balance columns of the worksheet for Dixon Company at June 30, 2017, are as follows. Other data: 1. A physical count reveals $500 of supplies on hand. 2. $100 of the unearned revenue is...
-
Under U.S. GAAP, an asset that is impaired should be written down in value, with an equivalent loss taken on the income statement. Discuss how you would determine whether an assets value was impaired...
-
Charles Choi was the owner/ operator of a grocery store in California called Genes Modern Market. In addition to the sale of normal grocery items, the store cashed payroll, personal, and third- party...
-
Suppose that in July 2 0 2 2 , Nike Inc. had EPS of $ 2 . 3 6 and a book value of equity of $ 9 . 1 6 per share. a . Using the average P / E multiple from the table here, , estimate Nike's share...
-
Which of the following reactions is (are) product-favored at equilibrium? (a) Zn(s) + 1(s) Zn+ (aq) + 2 I (aq) (b) 2 Cl (aq) + (s) Cl(g) + 2 [ (aq) (c) 2 Nat (aq) + 2 Cl (aq) 2 Na(s) + Cl(g) (d) 2...
-
You want to set up a series of voltaic cells with specific cell potentials. A Zn 2+ (aq, 1.0 M) | Zn(s) half-cell is in one compartment. Identify several half-cells that you could use so that the...
-
This problem is based on a recent annual report of the City of Tucson. Dates have been changed. 1. The MD&A reports that the city's general obligation bonds were rated AA-by Standard & Poor's. What...
-
Define and apply the following: Scarcity Needs Wants Microeconomics Four factors of production
-
Interactive Cooking or Beverage Workshop: Organize and conduct an interactive cooking or beverage workshop, integrating culinary theoretical knowledge with practical application. Design a workshop...
-
The manager of the company is looking to expand their business overseas. The expected cash flows of the project are given below. Cash flows Year $6,000 2 $ 33,600 4 $99,000 4 The interest rate over...
-
Four grams of fragrance are required for each bottle of Floral, a very popular perfume made by a small company in Lowell. The cost of the fragrance is $ 1 . 5 0 per gram. Budgeted production of...
-
A company reports the following: Line Item Description Amount Net income $ 1 0 0 , 8 7 0 Preferred dividends $ 7 , 4 7 0 Shares of common stock outstanding 2 0 , 0 0 0 Market price per share of...
-
Suppose a firm is equally likely to earn $2 million this year or lose $3 million. The firm faces a tax rate of 40% on each dollar of taxable income, and the firm pays no taxes on losses. In this...
-
A random sample of 10 houses heated with natural gas in a particular area, is selected, and the amount of gas (in therms) used during the month of January is determined for each house. The resulting...
-
(a) Implement the function F 1 = A'BC + B'C + ABC using an FPGA with programmable logic blocks consisting of 4-to-1 multiplexers. Assume inputs and their complements are available. (b) Implement the...
-
(a) In which applications should a designer use a CPLD rather than an FPGA? (b) In which applications should a designer use an MPGA rather than an FPGA? (c) In which applications should a designer...
-
(a) What is the difference between a traditional gate array and an FPGA? (b) What are the different types of FPGAs based on architecture (organization)? (c) What are the different programming...
-
1 2 3 Your firm is contemplating the purchase of a new $535,000 computer-based order entry system. The system will be depreciated straight-line to zero over its five-year life. It will be worth...
-
True/ false questions with explanations for the following statement 1) The expected return of zero beta security is smaller than risk free rate. 2) According to CAPM, the higher the variance, the...
-
The MMM Company has just paid a cash dividend of $9 per share. Investors require a 16 percent return from investments such as this. The dividend is expected to grow at a steady 8 percent per year....

Study smarter with the SolutionInn App


