A cell is constructed using the following half-reactions: (a) What reactions should be observed at the anode
Question:
A cell is constructed using the following half-reactions: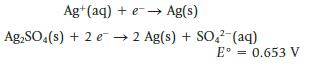
(a) What reactions should be observed at the anode and cathode?
(b) Calculate the solubility product constant, Ksp, for Ag2SO4.
Transcribed Image Text:
Ag+ (aq) + e → Ag(s) Ag₂SO4(s) + 2 e 2 Ag(s) + SO4²- (aq) E° = 0.653 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a The halfreaction with the more positive standard potential will occur at the cathode Therefore the ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A voltaic cell is constructed using the reaction (a) Write equations for the oxidation and reduction half-reactions. (b) Which half-reaction occurs in the anode compartment, and which occurs in the...
-
A potential of 0.142 V is recorded (under standard conditions) for a voltaic cell constructed using the following half reactions: (a) What is the standard reduction potential for the anode reaction?...
-
A voltaic cell is constructed using the reaction of chromium metal and iron(II) ions. Complete the following sentences: Electrons in the external circuit flow from the _______ electrode to the...
-
The accounting records of Shinault Inc. show the following data for 2017 (its first year of operations). 1. Life insurance expense on officers was $9,000. 2. Equipment was acquired in early January...
-
Dayna Moore, CEO of Layton Transmissions, sat dejected in her chair after reviewing the 2011 first-quarter financial reports on one of the companys core products: a standard, five-speed transmission...
-
Worksheet data for Savaglia Company are presented In Exercise, In Exercise, Instructions (a) Journalize the closing entries at April 30. (b) Post the closing entries to Income Summary and Owners...
-
Under U.S. GAAP, research and development costs are expensed as incurred; however, in the natural resource industry, the cost of finding and developing natural resources may be capitalized to the...
-
Refer to the Lufthansa example on asset depreciation estimates. What adjustments would be required if Lufthansas aircraft depreciation were computed using an average life of 25 years and salvage...
-
You have an opportunity to invest $50,000 now in return for $60,500 in one year. If your cost of capital is 7.6%, what is the NPV of this investment? The NPV will be $ (Round to the nearest cent.)
-
Which of the following reactions is (are) product-favored at equilibrium? (a) Zn(s) + 1(s) Zn+ (aq) + 2 I (aq) (b) 2 Cl (aq) + (s) Cl(g) + 2 [ (aq) (c) 2 Nat (aq) + 2 Cl (aq) 2 Na(s) + Cl(g) (d) 2...
-
You want to set up a series of voltaic cells with specific cell potentials. A Zn 2+ (aq, 1.0 M) | Zn(s) half-cell is in one compartment. Identify several half-cells that you could use so that the...
-
From the information of M Seaview, shown in figure 8.12, prepare the bank reconciliation statement and cash balance summary, then post the cash journals to the bank account in the general ledger to...
-
When Bruce Reid was hired as Blake Memorial Hospitals new CEO, the mandate had been clear: improve the quality of care, and set the financial house in order. As Reid struggled to finalize his budget...
-
Equivalent Units: Weighted Average Method The following data are for four independent process-costing departments. Inputs are added continuously. A B C D Beginning inventory 4,000 4,000 41,000...
-
A company has its share currently selling at $16.02 and pays dividends annually. The company is expected to grow at a constant rate of 3 percent pa.. If the appropriate discount rate is 16 percent...
-
Company Express S. A . asks you to construct cash flows for following three (3) investment projects, containing following information: PROJECT 1 a. Sales (in ThCh$): - Year 1: 90,000 - Year 2: 55,000...
-
According to CDC estimates, more than 2.8million people in the United States are sickened each year with antibioticresistant infections, with at least 35,000 dying as a result. Antibiotic resistance...
-
Why would a taxpayer be willing to pay a lawyer to provide a written opinion to a third party of the tax treatment to be accorded a particular set of transactions?
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
A Mealy sequential circuit with four output variables is realized using a 22V10. What is the maximum number of input variables it can have? What is the maximum number of states? Can any Mealy circuit...
-
Show how the left shift register of Figure 2-41 could be implemented using a CPLD. Draw a diagram. Give the equations for the flip-flop D inputs. always @ (posedge CLK) begin if (CLR) else if (Ld)...
-
An N-bit bidirectional shift register has N parallel data inputs, N outputs, a left serial input (LSI), a right serial input (RSI), a clock input, and the following control signals: Load: Load the...
-
Complete business activity and instalment activity statements in Australia Business Activity & Instalment activity Statement Tasks Sparky's Electrical Wholesale is registered forGST and reports on a...
-
1.In response to a Covid-19 induced drop in business, the Walt Disney Company furloughed 100,000 workers in April 2020. However, the company still plans to pay a $1.5 billion dividend in July. Asked...
-
CK Ltd is a major producer of blank CDs. The company uses a flexible budget and standard costs to aid planning and control of its machining manufacturing operations. Its costing system for...

Study smarter with the SolutionInn App


