Sodium azide, NaN 3 , undergoes the reaction NaN 3 (s) 2Na(s) + 3N 2 (g).
Question:
Sodium azide, NaN3, undergoes the reaction NaN3(s) → 2Na(s) + 3N2(g). Because this reaction is very fast and produces nitrogen gas, NaN3 is used to inflate airplane escape chutes. Sodium azide can be produced through two reaction steps.
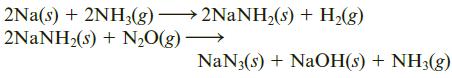
Starting with 1.0 kg of Na, 6.0 kg of NH3, and 1.0 kg of N2O, what is the maximum mass (kg) of sodium azide that can be produced?
Transcribed Image Text:
2Na(s) + 2NH3(g) 2NANH,(s) + H2(g) 2NANH2(s) + N,O(g) - NaN3(s) + NaOH(s) + NH3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
This problem is best approached by first combining the two steps given in the s...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
Starting with sodium azide as your source of nitrogen and using any other reagents of your choice, show how you would prepare each of the compounds in Problem 23.18. Compounds in 23.18 (a) (b) (c)...
-
What is the effect of a viscosity (competence) difference between strain markers and the matrix?
-
A bridge with a semielliptical arch spans a river as shown here. What is the clearance 6 ft from the riverbank? 14 ft 50 ft
-
Find the radiation power developed by a non-relativistic particle with charge e and mass m, moving along a circular orbit of radius R in the field of a stationary point charge q.
-
The annual report of Starbucks Corporation for the year ended September 29, 2013, included information on seven investments in which Starbucks generally held a 50 percent ownership interest. Thus...
-
Quigley Corporations trial balance at December 31, 2014, is presented below. All 2014 transactions have been recorded except for the items described below. Unrecorded transactions 1. On January 1,...
-
2. The Horace Corporation's inventory at December 31, 2017, was $330,000 based on a physical count priced at cost, and before any necessary adjustment for the following: Merchandise costing $38,000...
-
Create an ER model for each of the following descriptions: (a) A large organization has several parking lots, which are used by staff. (b) Each parking lot has a unique name, location, capacity, and...
-
Dinotrogen monoxide, commonly known as laughing gas, can be obtained by cautiously warming ammonium nitrate according to the equation NH 4 NO 3 (s) N 2 O(g) + 2H 2 O(g) If the reaction has a 75%...
-
A sample containing only boron and fluorine was decomposed yielding 4.75 mg of boron and 17.5 mL of fluorine gas (density = 1.43 g/L). What is the empirical formula of the sample compound?
-
Determine the best feasible solution among the following (feasible and infeasible) solutions of the Reddy Mikks model: (a) XI = 1, X2 = 4. (b) Xl = 2, X2 = 2. (c) XI = 3, x2 = 1.5. (d) XI = 2, X2 =...
-
What are the implications of adaptive radiation and convergent evolution for understanding species diversification, and how do these processes inform our knowledge of the evolutionary history and...
-
You have been requested to develop a digital advertising campaign for the Christmas campaign of the department store Harrods. Harrods Limited is a department store located on Brompton Road in...
-
Marigold Inc. is planning to invest in new manufacturing equipment to make a new garden tool. The garden tool is expected to generate additional annual sales of 8,000 units at $30 each. The new...
-
the npv is 27,942.61, the payback occurs in year 3.32, the discount payback occurs in year 4.38 and was rejected, the IRR is 20.77%, the PI is 0.093 and was rejected, and the firm wide WACC is 17.48....
-
Compare and contrast the shareholder theory and the stakeholder theory. Discuss which theory you agree with more and why. Give examples to support your discussion. (Best to give some law case related...
-
The spread between the yield on a three-year corporate bond and the yield on a similar risk-free bond is 50 basis points. The recovery rate is 30%. Estimate the average hazard rate per year over the...
-
What are the 5 Cs of marketing channel structure?
-
Hydrogen peroxide, H2O2, is a colorless liquid. A concentrated solution of it is used as a source of oxygen for rocket propellant fuels. Dilute aqueous solutions are used as a bleach. Analysis of a...
-
Nitric acid, HNO3, is a colorless, corrosive liquid used in the manufacture of nitrogen fertilizers and explosives. In an experiment to develop new explosives for mining operations, a sample...
-
Hydrogen cyanide, HCN, is a volatile, colorless liquid with the odor of certain fruit pits (such as peach and cherry pits). The compound is highly poisonous. How many molecules are there in 56 mg...
-
Which is true of adding a video to a Google Slides presentation? O you need to have the URL in order to add a video directly, otherwise, you have to download it to your computer first O you can...
-
Which statement about competency models is true? OThey help HR professionals ensure that all aspects of talent management are aligned with an organization's strategy. OThey identify and describe a...
-
Good time management includes always completing your easiest task first which will create a sense of accomplishment and motivate you to tackle next the most difficult task. True False

Study smarter with the SolutionInn App


