Consider the following liquid phase reactions: A plot of the concentrations in [mol/L], of A, B, and
Question:
Consider the following liquid phase reactions: ![]()
A plot of the concentrations in [mol/L], of A, B, and C in a constant-volume batch reactor operating isothermally at 500 K is shown in the following fi gure. Determine the values of K1 and K2 at 500 K.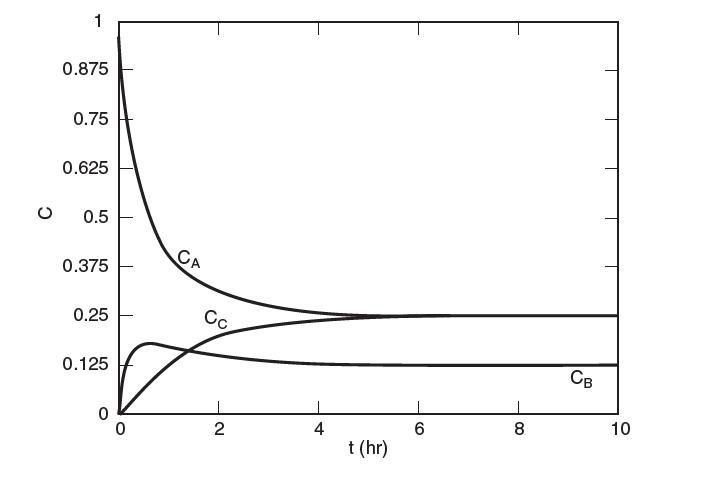
Transcribed Image Text:
2A B C
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
a. Example 8-1: Trambouze Reactions Wolfram 1. Fix k 2 at 0.015 and then describe how the selectivity varies with k 1 and k 3 . 2. Write a conclusion about your experiments. Polymath 3. What would...
-
The production of maleic anhydride by the air oxidation of benzene was studied using a vanadium pentoxide catalyst [Chem Eng Sci, 43, 1051 (1988)]. The reactions that occur are C6H6 + 9/ 2 O2 C4H2O3...
-
When a cosmetic manufacturer tests the market to determine how many women will buy eyeliner that has been tested for safety without subjecting animals to injury, is it involved in a descriptive...
-
Why might increasing taxes as a fiscal policy be a more difficult policy than the use of monetary policy to slow down an economy experiencing inflation? A. The legislative process works quickly. B....
-
Find the values of the trigonometric functions. Round off results. csc 82.4
-
Use the data for Albin Company in Problem 5-4B to complete the following requirements: Required 1. Prepare closing entries as of March 31, 2005 (the perpetual inventory system is used). Analysis...
-
Research and then discuss the implications of financing through debt as they compare to financing through equity. What are the pros and cons of each method? Which method would you use to raise...
-
Required information [The following information applies to the questions displayed below.] Golden Corporation's current year income statement, comparative balance sheets, and additional information...
-
The standard half-cell potential for the reduction of water to form hydroxyl ion in Table 9.1 is reported as -0.828 V. If instead we write the reaction: what should we use for the half-cell...
-
Which of the following conditions would you use if you needed to develop an industrial process to produce ethanol from acetylene? Explain. (a) 25C and 1 bar. (b) 250C and 1 bar. (c) 25C and 150 bar....
-
What percentage increase in wavelength leads to a 75% loss of photon energy in a photon-free electron collision?
-
The blade of a fan rotates with angular velocity given by the equation: w(t) = (5-0.8t 2 ) k rad/s). a) Find the angular displacement in the interval t=0s at=3s. b) If it is known that the length of...
-
This is a real project and is typical of the type of projects that are done by International Development Agencies such as the UNDP (United Nations Development Program), the Commonwealth Secretariat,...
-
4. Write a program to: (a) Display a "?" (b) Read two decimal digits whose sum is less than 10 (c) Display them and their sum on the next line, with an appropriate message.
-
Portsmouth Company makes upholstered furniture. Its only variable cost is direct materials. The demand for the company's products far exceeds its manufacturing capacity. The bottleneck (or...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Sales Variable manufacturing and...
-
Presented below is a partial trial balance for the Kansas Instruments Corporation at December 31, 2011. Additional information: 1. The note receivable, along with any accrued interest, is due on...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Explain whether you would expect KBr or CH3Br to have the higher melting point.
-
Which of these isomers would you expect to have the higher boiling point: Explain? CH 3 CH 2 CH 2 OH or CH 3 CH 2 OCH 3
-
Which of these compounds would you expect to be more soluble in water? Explain? CHCHCHCHCOH or CH3CHCHCHCHCOH
-
To seek innocent spouse relief, separation of liability relief, or equitable relief, which form should a taxpayer submit to the IRS? Form 843. Form 2210. Form 8379. Form 8857
-
SANDHILL MANAGEMENT CONSULTANTS Consolidated Income and Retained Earnings Statementfor the Year Ended December 31, 2024 Revenues 76,700 Operating Expenses including Depreciation of 2,100 francs...
-
You look at another report and find wages for the bar for another period were $11,421. At that time, you recall you operated staff in the bar on a 9% of sales basis. What sales did you need to make,...

Study smarter with the SolutionInn App


