Which of the following conditions would you use if you needed to develop an industrial process to
Question:
Which of the following conditions would you use if you needed to develop an industrial process to produce ethanol from acetylene? Explain.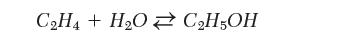
(a) 25°C and 1 bar.
(b) 250°C and 1 bar.
(c) 25°C and 150 bar.
(d) 250°C and 150 bar.
Transcribed Image Text:
CH4+ HO CH5OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Acetylene C2H2 is a different molecule than ethylen...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
If Figure 1 shows our production possibilities frontier during the Great Depression, at which point were we operating? a) point A b) point B c) point C d) point D A Figure 1 B C D
-
Darling Corporation issued 200,000 shares of $20 par value, 5% preferred stock on January 1, 2019, for $4,500,000. In December 2021, Darling declared its first dividend of $800,000. Part 1 Prepare...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Consider a group of 12 employees of whom five are in management and seven do clerical work. Select at random a sample of size 4. What is the probability that there will be one manager in this sample?
-
1. Why might federal spending on roads, waterways, or national security be less subject to direct expenditure offsets than spending on health care or education? 2. What might account for the fact...
-
Each given point is on the terminal side of an angle. Show that each of the given functions is the same for each point. (40, 30), (56, 42), (36, 27), csc and cos
-
Albin Companys adjusted trial balance on March 31, 2005, its fiscal year-end, follows: Required 1. Calculate the companys net sales for the year. 2. Calculate the companys total cost of merchandise...
-
The cost of materials transferred into the Filling Department of Savannah Lotion Company is $ 13,300, including $ 5,000 from the Blending Department and $ 8,300 from the materials storeroom. The...
-
Based on Georges assumptions that all of the remaining supervisors costs are split 65% to accounts payable and 35% to bank reconciliation, and all incremental ongoing technology costs and postage...
-
Consider the following liquid phase reactions: A plot of the concentrations in [mol/L], of A, B, and C in a constant-volume batch reactor operating isothermally at 500 K is shown in the following fi...
-
Your colleague reports that the conversion to n-butane from the gas phase hydrogenation reaction of 1-butene increases as temperature increases: Is this possible? Explain. C4Hs(g) + H(g) C4H0(g)
-
In teams of three or four, assume that you are the co- owners of a five-star restaurant located in the heart of a large city in the United States. First, draft a clause in a contract that might serve...
-
The first production department of Stone Incorporated reports the following for April. Direct Materials Conversion Units Beginning work in process inventory 80,000 Percent Complete 60% Percent...
-
What are the primary international frameworks governing banking regulations, such as Basel III, and how do they aim to mitigate systemic risk in the global financial system? Explain the challenges...
-
20Y3 20Y2 20Y1 Accounts receivable, end of year $705,000 $630,000 $595,000 Sales on account 5,609,000 4,602,000 This information has been collected in the Microsoft Excel Online file. Open the...
-
What is the difference between a stock and a bond? What is the purpose of a stock exchange? What is diversification in investing? What is a mutual fund? What is a hedge fund? What is the difference...
-
Byrnes Company accumulates the following data concerning a mixed cost, using units produced as the activity level. Units Produced Total Cost March 9,800 $14,740 April 8,500 13,250 May 7,000 11,100...
-
The following is a December 31, 2011, post-closing trial balance for the Valley Pump Corporation. Additional information: 1. The $120,000 balance in the land account consists of $100,000 for the cost...
-
1. Use these cost, revenue, and probability estimates along with the decision tree to identify the best decision strategy for Trendy's Pies. 2. Suppose that Trendy is concerned about her probability...
-
Which functional group is present in each of thesecompounds? a) CHCHCHCHOCH, ) CHCH,CH,CO,H Careful: how are the O's bonded to the C? e) CHCOCH, b) d) f) OH 0 CNH, H
-
Convert the following structures to skeletal structures: OH a) CHCH,CH,CHCH,CH, b) HC d) || HC OH CH CH HC. H CH3 CH CH H H H H CH f) CHCHCH-CHC-CH CH CH 0 11 c) CH3CCHCHCHCI || C CH3 e) CHC-CHCHCHCH...
-
Convert the following shorthand representations to structures showing all of the atoms, bonds, and unshared electron pairs: d) COOH COH b) OH de c) f) H CN Ni
-
SANDHILL MANAGEMENT CONSULTANTS Consolidated Income and Retained Earnings Statementfor the Year Ended December 31, 2024 Revenues 76,700 Operating Expenses including Depreciation of 2,100 francs...
-
In job-order costing system Question 1 oWhat is correct about job-order system? Question 2 options: all day to day expenses related to manufacturing of product by the firm are treated as factory...
-
What is the main purpose of a photographer?Select one:a.To write a script for the show.b.To finance and handle the production and cost needed in order to publish a print material.c.To capture images...

Study smarter with the SolutionInn App


