Determine the model parameters for the two-suffi x and the three-suffi x Margules equations using the liquidvapor
Question:
Determine the model parameters for the two-suffi x and the three-suffi x Margules equations using the liquid–vapor equilibrium data for a binary system of benzene (1)–cyclohexane (2) at 10°C. Mole fractions of liquid vs. total pressure are reported in Table E8.9A.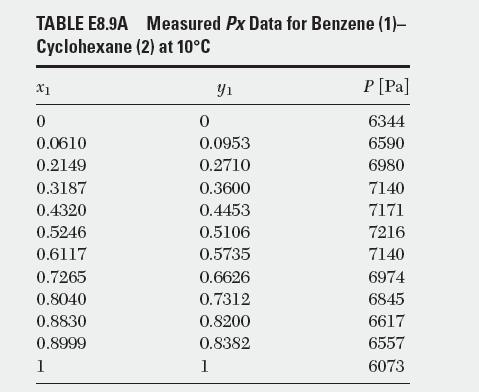
Transcribed Image Text:
TABLE E8.9A Cyclohexane X1 0 0.0610 0.2149 0.3187 0.4320 0.5246 0.6117 0.7265 0.8040 0.8830 0.8999 1 Measured Px Data for Benzene (1)- (2) at 10C Y 0 0.0953 0.2710 0.3600 0.4453 0.5106 0.5735 0.6626 0.7312 0.8200 0.8382 1 P[Pa] 6344 6590 6980 7140 7171 7216 7140 6974 6845 6617 6557 6073
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The excess Gibbs energy can be written g RTx ln y x ln y RT x In 1 RT 2 la 5 x la 5 In XPat where we ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Calculate the three-suffi x Margules parameters, A and B, for the system of Example 8.9. Example 8.9 Liquidvapor equilibrium data have been collected for a binary system of benzene (1)cyclohexane (2)...
-
Liquidvapor equilibrium data have been collected for a binary system of benzene (1)cyclohexane (2) at 10C. Mole fraction of liquid and vapor vs. total pressure are reported in Table E8.9A.5 From...
-
A binary system of species 1 and 2 consists of vapor and liquid phases in equilibrium at temperature T for which In y1 = 1.8x22 In y2 = 1.821. P1sat = 1.24 bar P2sat = 0.89 bar. Assuming the...
-
How do standard costs illustrate the trade-off between decision making and control?
-
When preparing a statement of cash flows wider the indirect method, an increase in ending accounts receivable over beginning accounts receivable will result in an adjustment to net income in the...
-
A rectangular grazing range with an area of 8mi 2 is to be fenced. Express the length l of the field as a function of its width w. What are the domain and range of l = f (w)?
-
Referring back to the information in the previous question, what would the revised balance on the suspense account be? Data From Previous Question: A business had a suspense account balance of 580...
-
Dudley Savings Bank wishes to take a position in Treasury bond futures contracts, which currently have a quote of 105-100. Dudley Savings thinks interest rates will go down over the period of...
-
- Linkage among the rations between Enel group and Engine group, from 2017 To 2021 ? - Strenghths and weaknesses between Enel group and Engine group, from 2017 To 2021 ?
-
Calculate the solubility of O2 from air in the atmosphere in equilibrium with liquid H2O at 25C. Report the answer in mole fraction and in molarity.
-
Show that the composition of the vapor and liquid phases must be equal at an azeotrope.
-
A data set is formed by recording the sums on 200 rolls of a pair of fair dice. (A) What would you expect the mean of the data set to be? The median? (B) Form such a data set by using a graphing...
-
A large handful of identical gemstones, totaling 195 carats (one carat has a mass of 200mg) at a temperature 73.2 degrees C, is placed in a calorimeter filled with 0.59 kg of water at 13.6 degrees C....
-
V. Describe why networks are evolving towards Spine-Leaf Architecture Design. Differentiate 3-tier and Spine-Leaf.
-
Explain how shorter SID becomes a contributing factor for distortion once it is already occurring due to misalignment.
-
A 95% confidence interval is (54.5, 57.5) based on a sample size of 25. What is the sample standard deviation?
-
A company estimates the following manufacturing costs at the beginning of the period: direct labor, $532,000; direct materials, $196,000; and factory overhead, $123,000. Required: 1. Compute its...
-
Define the terms working capital, current ratio, and acid-test ratio (or quick ratio).
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
Cules son las ventajas de las redes sociales de pago? Selecciona todas las opciones que correspondan. 1 punto Tienen un alcance ms rpido Se dirigen a clientes relevantes Mejoran el servicio al...
-
Assume that on June 1, 20Y2, Peak Company purchases 3,000 sharesof Antares Company common stock at $59.90 per share plus abrokerage commission of $325. The entry to record the purchase ofthe stock...
-
Lionel is 52 years of age and is a single parent. He has two dependent children living with him, twin daughters, Clara and Joanne, aged 17 years. Clara has no income, but Joanne is in receipt of a...

Study smarter with the SolutionInn App


