Assign configuration to the following substrate, and show the stereochemistry and identity of the product you would
Question:
Assign configuration to the following substrate, and show the stereochemistry and identity of the product you would obtain by SN1 reaction with H2O (reddish brown = Br):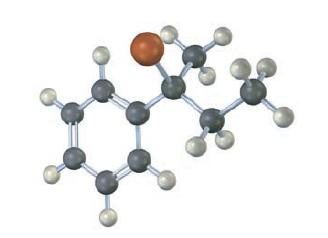
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To assign the configuration to the given substrate and predict the product obtained by SN1 reaction ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Assign configuration to the following substrate, and show the stereo chemistry and identity of the product you would obtain by SN1 reaction with water (reddish brown =Br):
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
Assign R or S configuration to the following molecule, write the product you would expect from SN2 reaction with NaCN, and assign R or S configuration to the product (yellow-green =Cl):
-
The passage indicates that the late 1850s Democrats: F. Were all Southern slaveholders who wanted to expand slavery into the territories. G. Used legislation in the early 1850s to support their...
-
In the aftermath of a hurricane, an entrepreneur took a one- month leave of absence (without pay) from her $ 5,000 per month job in order to operate a kiosk that sold fresh drinking water. During the...
-
The Plated Pixie Company produces meal kits. Each week, it ships thousands of meal kits. Each meal kit includes a variety of foods (direct materials). Company managers have the following...
-
Poplar Outdoor Corporation owns 60 percent of the voting stock of Sugg Australia. Date-of-acquisition information is as follows: Acquisition cost: \($39.5\) million Fair value of the noncontrolling...
-
Pro Tire manufactures tires for all- terrain vehicles. Pro Tire uses job costing and has a perpetual inventory system. On November 22, Pro Tire received an order for 190 TX tires from ATV Corporation...
-
Dexter Mills issued 10-year bonds a year ago at a coupon rate of13.2 percent. The bonds make semiannual payments. Theyield-to-maturity on these bonds is 9.6 percent. Assume the parvalue is 1,000 US 2...
-
Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? (a) Br CH3 | | CH3CH2CHCHCH3 (b) CH3 CI CH3 | | CH3CHCH2-C-CHCH3 CH3...
-
Assign R or S configuration to the following molecule, write the product you would expect from S N 2 reaction with NaCN, and assign R or S configuration to the product (red = O, yellow-green = Cl):
-
If you were a debtor in financial difficulty who thought that your creditors were likely to seek a bankruptcy order against you in the next few months, could you protect some of your assets against...
-
ACME Enterprise currently sells candies for $250 which includes a mark-up of 25%. Each order is placed by a single individual that charges an average rate of $40 per hour. To file the paperwork as...
-
Use Marshall and Swift and the format in Sample Appraisal 3 to apply the Cost Approach to 100 Woodbridge Center Drive, Woodbridge,...
-
You have isolated DNA from three different cell types of an organism, determined the relative DNA content for each type, and plotted the results on the graph shown in the Figure Below. Refer to the...
-
Bob is 37 years old and is an account manager with a large telecom company.He's been dragging his feet with his retirement plan, opting instead for expensive trips to Vegas and Aspen.He's decided...
-
Entity A writes a single policy for a P100,000 premium and expects claims to be made of P60,000 in 2013. At the time of writing the policy, there are commission costs of P20,000. Assume a discount...
-
How can special government agencies help promote trade?
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
When (R)-3-bromo-2, 3-dimethylpentane is treated with sodium hydroxide, four different alkenes are formed. Draw all four products, and rank them in terms of stability. Which product do you expect to...
-
When 3-bromo-2, 4-dimethylpentane is treated with sodium hydroxide, only one alkene is formed. Draw the product and explain why this reaction has only one regiochemical outcome.
-
Predict the major product for each of the following E2 reactions: a. b. c. d. NaOH Br NaOH Br
-
Indirect costs Blank______. Multiple choice question. are not easily traced to products or services must be assigned using volume-based cost systems are only related to manufacturing activities are...
-
Foreign income earned can be converted to Canadian dollars using the Bank of Canada exchange rate in effect on the day that the taxpayer received the income. Question 11 options: True False Question...
-
Under Sarbanes-Oxley, the audit committee is expected to Blank______. (Check all that apply.) Multiple select question. establish an internal audit function establish and implement internal controls...

Study smarter with the SolutionInn App


