-Glucose has the following structure. Identify the chirality centers in -glucose, and tell how many stereoisomers of...
Question:
β-Glucose has the following structure. Identify the chirality centers in β-glucose, and tell how many stereoisomers of glucose are possible.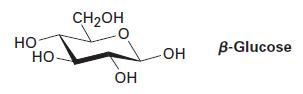
Transcribed Image Text:
НО НО CH₂OH ОН -ОН OH B-Glucose
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
In glucose the structure is HO CH2OHCOH OH glucose has four ch...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify all chirality centers in each of the following compounds: a. b. c. d. e. f. . Ascorbic acid (Vitamin C) Vitamin D3
-
The Fischer projection formula for glucose (blood sugar, Sec. 16.4) is Altogether, how many stereoisomers of this sugar are possible? CH= O HOH HOH CH OH glucose
-
Extract of Ephedra sinica, a Chinese herbal treatment for asthma, contains the compound ephedrine, which dilates the air passages of the lungs. The naturally occurring stereoisomer is levorotatory...
-
Two wheels A and B have masses m A and m B and radii of gyration about their central vertical axes of k A and k B respectively. If they are freely rotating in the same direction at A and about the...
-
A firm produces output according to a production function Q = F (K, L) = min {4K, 8L}. a. How much output is produced when K = 2 and L = 3? b. If the wage rate is $ 60 per hour and the rental rate on...
-
Refer to the data in E11-26A. Requirements: 1. Make the journal entries to record the purchase and use of direct materials. 2. Make the journal entries to record the direct labor. Data From E11-26A:-...
-
Pierre Corporation acquired 75 percent of Selene Corporation's common stock for \($20,100,000\) on January 2, 2014. The estimated fair value of the noncontrolling interest was \($5,900,000.\)...
-
The records of Ellens Boutique report the following data for the month of April. Instructions Compute the ending inventory by the conventional retail inventorymethod. $99,000 Purchases (at cost)...
-
. A bond issued by Coca Cola Co. matures on 19 January 2093, andhas the coupon rate of 7.375%. Given that the bond pays semi-annualcoupon payments and recently traded at 121.971 (percent per parval 2...
-
How would you prepare the following alkyl halides from the appropriate alcohols? (a) CI CH3CCH3 CH3 CH3 CH3CHCHCHCH3 (b) Br (c) CH3 T BrCHCHCHCHCHCH3 (d) CH3 CI I I CH3CHCHCHCCH3 CH3
-
Write representative structures for the following: (a) A fat (b) A vegetable oil (c) A steroid
-
Use implicit differentiation to find dy/dx. x + y 2 = sin y
-
A firm buys capital equipment for $1000 in Year 0. The annual depreciation, starting in Year 1, is $200. In Year 3, immediately after taking the third depreciation, the firm sells the equipment for...
-
Consider a bond with a 6% coupon rate that matures in 6 years. Coupons are paid annually, and the face value is $1,000. If the bond is currently selling for $952.00, what is the yield to maturity?
-
Suppose that TechnoTCL is considering a new project. They are trying to determine the required rate of return for their debt and equity holders. See the information below: A 6 . 5 % ?percent annual...
-
Consider an investor who has purchased a put option on XYZ Corporation with a strike price of $50. The investor paid a premium of $2 for the put option. What will be the investor's return (in...
-
Q1. You believe stock price by year end will have the following multinomial distribution: Price Probability 60 10% 80 20% 100 40% 120 20% 140 10% Q1a. What should be the stock price TODAY? Q1b. what...
-
Describe the national competitive advantage theory. What is an advanced factor?
-
Calculate the number of neutrons of 239Pu.
-
The following two compounds are both secondary alkyl halides, but they undergo E2 reactions at different rates. The first compound reacts more rapidly than the second compound. Explain. Br Br
-
The following three reactions are similar, differing only in the configuration of the substrate. One of these reactions is very fast, one is very slow, and the other does not occur at all. Identify...
-
1-Bromobicyclo [2.2.2] octane does not undergo an E2 reaction when treated with a strong base. Explain why not.
-
1. Z-Mart had sales of $354,700. Its cost of goods sold was $143,400. What is the gross profit? A) $211,300. B) $231,500. C) $354,700. D) $143,400. E) 40%. 2. Z-Mart had sales of $569,300. Its gross...
-
Interest paid on the principal and also on previously earned interest, assuming that the interest is left in the account, is called _____________________. Group of answer choices Compound Interest...
-
A standard is a unit amount and a budget is a total amount. Group of answer choices True False

Study smarter with the SolutionInn App


