How would you prepare the following alkyl halides from the appropriate alcohols? (a) CI CH3CCH3 CH3 CH3
Question:
How would you prepare the following alkyl halides from the appropriate alcohols?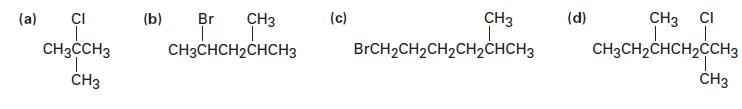
Transcribed Image Text:
(a) CI CH3CCH3 CH3 CH3 ī CH3CHCH₂CHCH3 (b) Br (c) CH3 T BrCH₂CH₂CH₂CH₂CHCH3 (d) CH3 CI I I CH3CH₂CHCH₂CCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
To prepare the given alkyl halides from the appropriate alcohols we need to perform a substitution r...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you prepare the following alkyl halides from the correspondingalcohols? CH (a) CI (b) Br CH CHH2CH CH (c) CI H (d) BrCH2CH2CH2CHCH CHCH2H2cH3 CH
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
How many alkyl halides can be obtained from monochlorination of the following alkanes? Neglect stereoisomers. (a) CH3CH2CH2CH2CH3 (b) (c) (d) (e) (f) (g) (h) (i) CH3 H3 CH CHCH2CH2CHCH3 , CH3 CH3...
-
For Problem 511, complete the design of a pair of helical gears to operate under the stated conditions. Specify the geometry of the gears and the material and its heat treatment. Assume that the...
-
A firm produces output according to the production function Q = F (K, L) = 4K + 8L. a. How much output is produced when K = 2 and L = 3? b. If the wage rate is $ 60 per hour and the rental rate on...
-
Melford Containers currently uses a recycled plastic to make bottles for the food industry. Current bottle production information: The cost and time standards per batch of 10,000 bottles are as...
-
Pentland Shoe Company acquired 75 percent of Sketchers Inc.'s common stock on January 2, 2014 for \($180\) million. The fair value of the 25 percent noncontrolling interest was \($50\) million at the...
-
R. Herbert of Crestview Manufacturing Co. is paid at the rate of $20 an hour for an eight-hour day, with time-and-a-half for overtime and double-time for Sundays and holidays. Regular employment is...
-
How long will it take money toquadrupleif it is invested at9?%compoundedsemiannually?6.4%compounded? continuously? 2 answers
-
What substitution products would you expect to obtain from the following reactions? (a) (c) Br T CH3CHCHCH3 + LII CHBr + NaCN ? (b) CH3 T CH3CHCHCI + HS ?
-
-Glucose has the following structure. Identify the chirality centers in -glucose, and tell how many stereoisomers of glucose are possible. CHOH - OH B-Glucose
-
The following data relate to Emporium Ltd. A dditional information 1. Dividends were paid during the year. 2. The increases in investments and machinery were from cash purchases. 3. The increases in...
-
1 - The management of KADJI expects to receive cash revenues of $120m, $180m and $300m four, five and six years from now, respectively.KADJI can earn an annual return of 5%, compounded annually, on...
-
The existing 10 year, 6% bonds are trading in the market at $900. The corporate tax rate is 32%. Calculate the interest rate of the new bonds. What is the after-tax interest rate for the new bonds?
-
KatyCo. is approached by a new customer to fulfill a large one-time-only special order for a product similar to one offered to domestic customers.KatyCo. has excess capacity. The following per unit...
-
Elena just bought a condo in downtown Vancouver, and the purchase price is $750,000. For this condo purchase, she used her savings to pay for the down payment of 30% of the purchase amount and...
-
3. Edwards plc manufactures silver cutlery which requires two key processes, Melting and Moulding, the output of Melting passing forward to Moulding. At the start of March 2021, 300 units were 20%...
-
What are the four elements and two influential factors of the Porter Diamond?
-
Define the essential properties of the following types of operating systems: a. Batch b. Interactive c. Time sharing d. Real time e. Network f. Parallel g. Distributed h. Clustered i. Handheld
-
Equation (3.38), C P = C V + TV ( 2 /). links C P and C V with and . Use this equation to evaluate C P C V for an ideal gas.
-
Draw the carbocation intermediate that would be formed if each of the following substrates participated in a stepwise elimination process (E1). In each case, identify the intermediate carbocation as...
-
Draw the transition state for the reaction between tertbutyl chloride and sodium hydroxide.
-
2. Method 2, Residual Income Model: Find or estimate the EPS growth rate. Find book value per share on the balance sheet. Use the discount rate k from part a or b above and estimate the value of your...
-
The underlying motivation for fraud is to Blank______. Multiple choice question. cover up an error in the financial statements achieve a gain over another party that would not otherwise exist...
-
ecure and confidential patient registration locations are considered to influence what type of organizational marketing efforts: Group of answer choices internal and controllable internal and...

Study smarter with the SolutionInn App


