How can you explain the observation that the SN2 reaction of (dibromomethyl)benzene with NaOH yields benzaldehyde rather
Question:
How can you explain the observation that the SN2 reaction of (dibromomethyl)benzene with NaOH yields benzaldehyde rather than (dihydroxymethyl)benzene?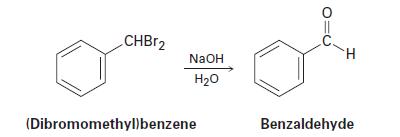
Transcribed Image Text:
CHBr2 NaOH H20 (Dibromomethylbenzene H Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The observation that the SN2 reaction of bromomethylbenzene with NaOH yields benzaldehyde rather than hydroxymethylbenzene can be explained by the mec...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The SN2 reaction of (dihromomethyl) benzene, C6H5CHBr2, with NaOH yields benzaldehyde rather than (dihydroxymethyl) benzene, C6H5CH (0H)2 Explain.
-
Draw the product(s) obtained by conjugate addition of the following reagents to cyclohex-2-enone: (a) H 2 O (b) NH 3 (c) CH 3 OH (d) CH 3 CH 2 SH How can you explain the observation that the S N 2...
-
Aromatic compounds such as benzene react with alkyl chlorides in the presence of A1C1 3 catalyst to yield alkylbenzenes. The reaction occurs through a carbocation intermediate, formed by reaction of...
-
Solve each system. If a system is inconsistent or has dependent equations, say so. -5x + 2y = -4 6x + 3y = -6
-
Some companies that use process costing simply assign the entire cost of production to those units completed and transferred during the month, even if some units remain in process at the end of the...
-
A sample contains \(3.0 \times 10^{22} \mathrm{Si}\) atoms and nothing else. (a) How many valence electrons does the sample contain? (b) How many of the valence electrons in the sample can conduct...
-
Which operations have the same precedence in order of operations?
-
Mark Corporation estimates its manufacturing overhead to be $90,000 and its direct labor costs to be $200,000 for year 1. The actual direct labor costs were $50,000 for Job 301, $75,000 for Job 302,...
-
Swifty Ltd. began business on January 1, 2019. At December 31, 2019, it had a $53,820 balance in the Deferred Tax Liability account that pertains to property, plant, and equipment acquired on July 1,...
-
Show the products that result from the reaction of phenylmagnesium bromide with the following reagents: (a) CH 2 O (b) Benzophenone (C 6 H 5 COC 6 H 5 ) (c) Pentan-3-one
-
Draw structures corresponding to the following names: (a) Bromoacetone (b) 3-Methylbutan-2-one (c) 3,5-Dinitrobenzaldehyde (d) 3,5-Dimethylcyclohexanone (e) 2,2,4,4-Tetramethylpentan-3-one (f)...
-
House Corporation has been operating profitably since its creation in 1959. At the beginning of 2011, House acquired a 70 percent ownership in Wilson Company. At the acquisition date, House prepared...
-
11. The Kamp family has twins, Rob and Rachel. Both Rob and Rachel graduated from college 2 years ago, and each is now sarning $50,000 per year. Rachel works in the retail industry, where the mean...
-
1.What is the break-even (NPV=0) annual incremental sales for the following investment? Initial investment at time t=0 is $2,400,000 Life of project is 12 years Initial investment depreciated to $0...
-
Until recently, Augean Cleaning Products sold its products on terms of net 64, with an average collection period of 79 days. In an attempt to induce customers to pay more promptly, it has changed its...
-
2. Suppose the S&P 500 index future is trading at 1,100. You would like to long 10 contracts? What is the total value of the stock index futures contracts? Use the multiplier of 250.
-
Convert 742 to Mayan Long Count (modified vigesimal). Give your answer using the base-sixty convention. If the representation is not unique, give the representation in which the digit in the 20's...
-
What is meant by the term joint venture? Identify four joint venture configurations.
-
Consider the sections of two circuits illustrated above. Select True or False for all statements.After connecting a and b to a battery, the voltage across R1 always equals the voltage across R2.Rcd...
-
Sigma bonds experience free rotation at room temperature: In contrast, Ï bonds do not experience free rotation. Explain. . cec - `H
-
Predict which of the following compounds is more acidic, and explain your choice. N- -N- -NH2 NH2
-
Consider the reaction below. The rate of this reaction is markedly increased if a small amount of sodium iodide is added to the reaction mixture. The sodium iodide is not consumed by the reaction and...
-
3 B B B B B B B B B ORTHOGRAPHIC NAME: PROJECTION CHAPTER 02 EXERCISE 11 DATE: VISUALIZATION & ENGINEERING DESIGN GRAPHICS WITH AUGMENTED REALITY SKETCH ALL NECESSARY VIEWS TO COMPLETELY DESCRIBE THE...
-
You are about to optimize the IP addressing table for a network with 5 different subnetworks and 20 hosts pers subnetwork. The Internet Service Provider gives you a class B network. The best...
-
%m-file to calculate eigenvalues and eigenvectors % inertia values in kg-m^2 J1=5.65; J2=15.6; J3=16.38; J4=20.45; J5=29.38; J6 .5*15818; % stiffness values in N-m/rad K1=1.7e06; K2 3.4e06;...

Study smarter with the SolutionInn App


